In anticipation of the upcoming 2024 Myofilament Meeting, April’s publication review covers recent advances in the myofilament field. From the effects of inotropic agents on cardiac myofilament Ca2+ sensitivity, protein kinase D’s role in cardiac homeostasis, and the contribution of specific myosin-binding protein C domains in muscle contraction, the following studies usher in a promising trend of exciting breakthroughs.
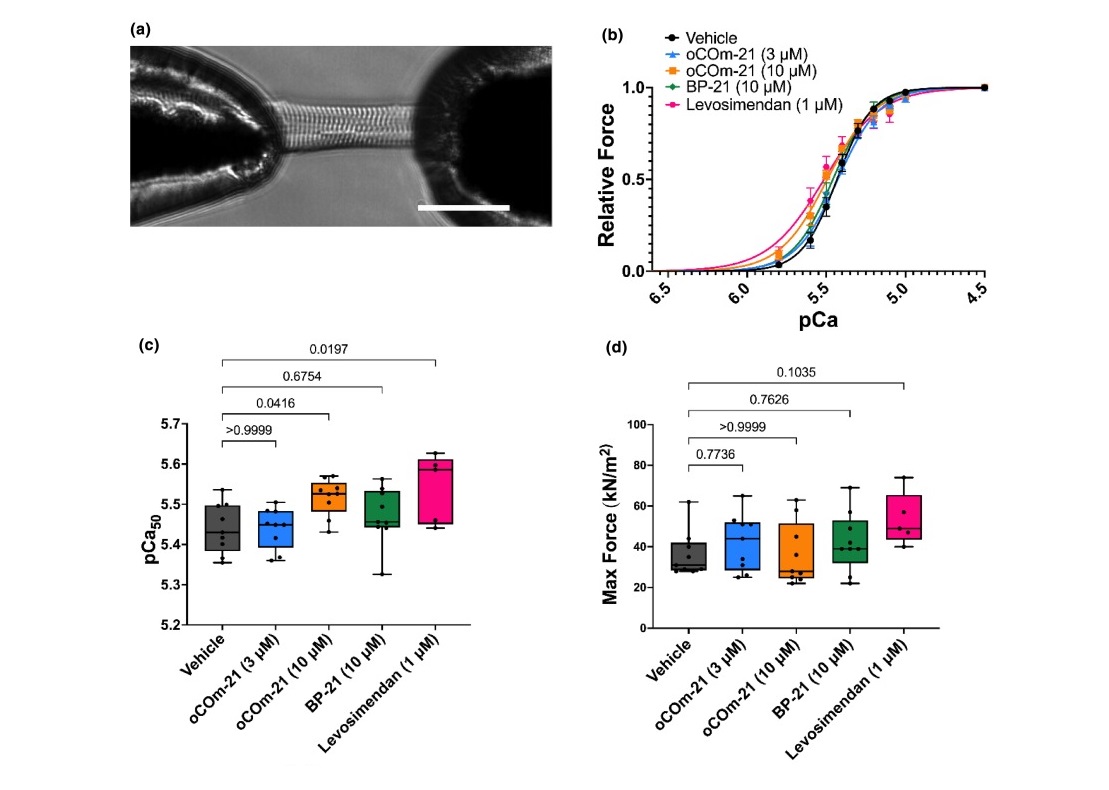
Featured image (adapted from ©Payne et al. (2024), licensed under CC BY 4.0 DEED) revealing oCOm-21 (10 μM) increases myofilament Ca2+ sensitivity. A) A permeabilized cardiomyocyte, glued to two micro-pins and connected to Aurora Scientific’s force transducer and piezoelectric motor arm at 20× magnification. Scale bar shown represents 50 μm. B) Force-pCa relationships of permeabilized cardiomyocytes across treatment and control groups. C) pCa50 values. D) Maximal active force values.
The carbon monoxide prodrug oCOm-21 increases Ca2+ sensitivity of the cardiac myofilament
Cardiopulmonary bypass procedures are performed to correct vascular defects in patients who have suffered from acute myocardial infarction, commonly known as a heart attack. Patients undergoing these procedures often receive inotropes to maintain or improve cardiac output and prevent complications such as low-output syndrome and organ dysfunction. While promising, traditional inotropes such as dobutamine can promote arrythmias due to their effect on intracellular calcium levels, underscoring the need for safer inotropic agents.
To address this, Payne et al. (2024) investigated the Ca2+ sensitizing effects of a carbon monoxide (CO)-releasing prodrug, oCOm-21, known to exhibit positive inotropic effects without any reported arrhythmogenicity. Left ventricular cardiomyocytes were isolated from male Sprague Dawley rats, permeabilized, and mounted to an inverted phase contrast microscope. Using silicone glue, the permeabilized cardiomyocytes were adhered to 100 μm pins connected to Aurora Scientific’s 406A force transducer and 322C high-speed length controller. Across the 8-well bath plate of Aurora Scientific’s 803B apparatus, the cardiomyocytes were immersed in a series of submaximal pCa solutions, and the 900B VSL camera was used to monitor sarcomere length.
Upon assessing pCa50, known as the Ca2+ concentration at which 50% of maximal force is produced, the researchers found that oCOm-21 significantly increased Ca2+ sensitivity of the myofilament compared to the vehicle and control conditions. Moreover, the oCOm-21 treatment revealed no change in myofilament phosphorylation compared to the vehicle, while further treatment with the heme scavenger, hemopexin, abolished the Ca2+ sensitizing effect of oCOm-21. Together, these findings suggest a heme dependent mechanism underlies oCOm-21-induced myofilament Ca2+ sensitivity in permeabilized cardiomyocytes, and provide a promising framework from which to explore its potential as a clinical inotropic agent.
Protein Kinase D plays a crucial role in maintaining cardiac homeostasis by regulating post-translational modifications of myofilament proteins
Protein Kinase D 1 (PKD1) is the main isoform of the PKD family of proteins. While PKDs are known to play a critical role in myocardial contraction, hypertrophy, and remodeling processes, the specific role of PKD1 on cardiomyocyte function during oxidative stress has not been explored. Herwig et al. (2024) therefore sought to investigate this by assessing myofilament properties and inflammation markers in cardiac tissue of cardiomyocyte-specific Prkd1 knockout (PKD1 cKO) mice.
Using Aurora Scientific’s 1600A system, single skinned cardiomyocytes from the left ventricles of cKO mice were attached with silicone adhesive between the 403A force transducer and 322C high speed length controller. After sarcomere length was adjusted, the cardiomyocytes were exposed to a series of different Ca2+ concentrations across the 8 wells of the 803B apparatus.
Interestingly, the permeabilized cardiomyocytes from PKD1 cKO mice exhibited increased passive stiffness, increased myofilament Ca2+ sensitivity, and reduced maximum Ca2+-activated tension. These effects were subsequently reversed upon treatment with reduced glutathione, suggesting that myofilament protein oxidation underlies cardiomyocyte dysfunction. Beyond these effects, PKD1 deficiency also resulted in increased oxidative stress and pro-inflammatory markers. Collectively, these findings underscore the importance of PKD1 in maintaining the balance of oxidative stress and inflammation to preserve cardiomyocyte function.
Myosin-binding protein C regulates the sarcomere lattice and stabilizes the OFF states of myosin heads
Myosin-binding protein C (MyBP-C) is a protein found in muscle cells, particularly in cardiac and skeletal muscle, where it plays a crucial role in regulating muscle contraction. Although mutations in the gene encoding MyBP-C are known to cause debilitating diseases such as hypertrophic cardiomyopathy, the specific roles of the middle- and N-terminal domains of MyBP-C (MyBP-CC1C7) have yet to be understood. Hessel et al. (2024) aimed to address this by performing both mechanical and small angle X-ray diffraction experiments in permeabilized skeletal muscle from SNOOPC2 mice.
To assess crossbridge kinetics and mechanics, permeabilized psoas fibers were attached between Aurora Scientific’s 315C high speed length controller and 403A force transducer with silicone. In their 1600A system set-up, the 803B temperature-controlled apparatus was regulated by the 825A thermocouple, located on the stage of an inverted microscope, and the 901D High-Speed VSL camera was used to monitor sarcomere length. As they went on to perform small-angle X-ray diffraction experiments, the muscle preps were hung between the 402A force transducer and 322 high-speed length controller, with force and length data collected on Aurora’s 600A: Real-Time Muscle Data Acquisition and Analysis System.
The researchers found that cleavage of MyBP-C lead to shorter thin filaments and reduced calcium sensitivity at shorter sarcomere lengths, while increasing cross-bridge kinetics across sarcomere lengths under submaximal activation. Additionally, X-ray structural analysis indicated a shift of myosin heads towards the ON state after cleavage, suggesting a key role for MyBP-CC1C7 domains in regulating contractile performance in muscle.
Conclusions
As we spring past the month of April, these studies by Payne et al. (2024), Herwig et al. (2024), and Hessel et al. (2024) shed light on the intricate mechanisms underlying muscle function, particularly in the context of myofilament regulation and contraction. From prodrugs to proteins, these insights ultimately deepen our understanding of muscle diseases, and spotlight potential therapeutic targets to improve muscle function.