803B: Permeabilized Myocyte Apparatus – Microscope Mountable
The 803B permeabilized myocyte test apparatus is engineered to be a reliable setup for testing the contractile properties of skinned myocytes. A large cell mounting area provides ample room for cell attachment. In addition, push-button controlled motorized stages simplify cell attachment to a high-speed length controller (315C, 322C) and precision force transducer (400A series).
A temperature controlled 8-well bath plate allows for seamless transition of the myocyte between varying calcium concentrations. Direct mounting on an inverted microscope stage allows for observation and sarcomere spacing detection. Additionally, an optional prism reticule can be included to provide accurate measurement of cell cross-sectional area.
Model 803B-XYZ provides motorized XYZ stages for the left and right side of the microscope along with push-button controls, damped rods and mounting platforms. This equipment arrangement provides the researcher with fast and accurate positioning control of both the length controller and the force transducer which is especially important while mounting the cell.
Temperature of the test plate can be controlled from 4 to 40°C within +/- 0.1°C using the supplied Aurora Scientific 825A thermoelectric controller. The 825A also contains a low noise thermocouple thermometer with both a digital readout and a voltage output. An Aurora Scientific model 826A Water Circulator is also included.
Components
825A Thermocouple Meter / TEC Controller:
Thermometer and TEC Controller controls bath temperature from 0 to 40°C
826A Water Circulator:
Water circulator that provides cooling for 801C, 802D, 803B apparatus. Includes water reservoir, pump and radiator with fan
Stories of Success
1600A – Turnkey Solution Aids Characterization of Cardiac Cell Mechanics in Heart Failure
CHALLENGE
Dr. Jonathan Kirk was working as a postdoctoral fellow in Dr. David Kass’ lab at Johns Hopkins University who had acquired his own custom built equipment for testing mechanics of heart failure in skinned cardiomyocytes. When Dr. Kirk secured a faculty position at Loyola University in Chicago, he did not have the means to fabricate his own custom apparatus and stages to characterize cardiomyocyte function. He also needed to find a way to integrate his own stepper motor for small-scale length changes in single cell preparations while measuring and monitoring sarcomere length.
SOLUTION
Aurora Scientific had worked closely with Dr. Kirk and Dr. Kass previously so we knew we would be able to provide him a complete solution for his new lab. The 1600A Permeabilized Myocyte System was the answer as it included our 803B 8-well skinned myocyte apparatus for various calcium concentrations, 3-axis motorized stages and a complete data acquisition and analysis system. Furthermore, Aurora Scientific provided our 400A series high-resolution force transducer and incorporated our video sarcomere length software (VSL) and camera for measuring and monitoring sarcomere spacing.
RESULTS
The permeabilized myocyte system was provided to Dr. Kirk, which was easily mounted on an inverted microscope and combined with motorized stages to deliver precise control of the transducer and motor for cell attachment and testing. Furthermore, the 8-well bath plate allowed for quick determination of force-pCA in these cells. Additionally, our data acquisition software and VSL enabled full integration of Dr. Kirk’s stepper motor for precise length changes, easily monitored using the VSL camera. The 1600A has provided Dr. Kirk with a system that will allow him to characterize mechanics of heart failure and discover the mechanisms involved, leading the way to the development of potentially ground-breaking treatments for humans with this condition.
Select References
- Alegre-Cebollada, Jorge et al. “S-glutathionylation of cryptic cysteines enhances titin elasticity by blocking protein folding.” Cell (2014) DOI: 10.1016/j.cell.2014.01.056
- Alegre-Cebollada, Jorge et al. “Large-Scale Modulation of Titin Elasticity by S-Glutathionylation of Cryptic Cysteines.” Cell (2014) DOI: 10.1016/j.cell.2014.01.056
- Kötter, Sebastian et al. “Human myocytes are protected from titin aggregation-induced stiffening by small heat shock proteins” The Journal of Cell Biology (2014) DOI: 10.1083/jcb.201306077
- Horiguchi, Hiroshi et al. “Fabrication and evaluation of reconstructed cardiac tissue and its application to bio-actuated microdevices.” IEEE Transactions on NanoBioscience (2009) DOI: 10.1109/TNB.2009.2035282
- Ahrens-Nicklas et al. “Disruption of cardiac thin filament assembly arising from a mutation in LMOD2: A novel mechanism of neonatal dilated cardiomyopathy” Science Advances (2019) DOI: 10.1126/sciadv.aax2066
- Hamdani, Nazha et al. “Deranged myofilament phosphorylation and function in experimental heart failure with preserved ejection fraction.” Cardiovascular Research (2013) DOI: 10.1093/cvr/cvs353
- DeWinter, Josine M. et al. “Troponin activator augments muscle force in nemaline myopathy patients with nebulin mutations.” Journal of Medical Genetics (2013) DOI: 10.1136/jmedgenet-2012-101470
- van der Pijl et al. “Titin-based mechanosensing modulates muscle hypertrophy” Journal of Cachexia, Sarcopenia and Muscle (2018) DOI: 10.1002/jcsm.12319
- Bennett et al. “RNA helicase, DDX27 regulates skeletal muscle growth and regeneration by modulation of translational processes” PLoS Genetics (2018) DOI: 10.1371/journal.pgen.1007226
- Garibaldi et al. “Novel ACTA1 mutation causes late-presenting nemaline myopathy with unusual dark cores” Neuromuscular Disorders (2021) DOI: 10.1016/j.nmd.2020.11.012
Related Products

315D/322D: High-Speed Length Controllers
The 315D/322D High-Speed Length Controllers give physiologists the ability to control and measure length of single cells, fibers and whole muscle with ease.
Learn More
400C: Force Transducers
The 400C series of force transducers is the next generation of our widely used 400A and 400B series. The 400C series enables contractile measurements from a variety of muscle types and sizes and are designed to meet the needs of muscle researchers.
Learn More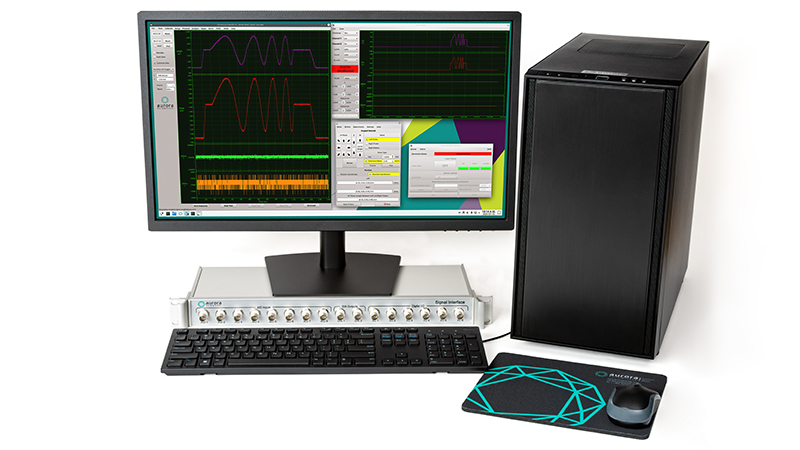
600A: Real-Time Muscle Data Acquisition and Analysis System
The 600A Digital Controller serves to integrate components and provide the researcher control of system operations, data collection and signal analysis.
Learn More
820A: Dual XYZ Motion Controller
Closed loop control of motorized stages to easily perform complex micro positioning with stunning precision
Learn More
825B: TEC Controller / Thermometer
Thermometer and TEC Controller controls bath temperature from 0 to 40°C
Learn More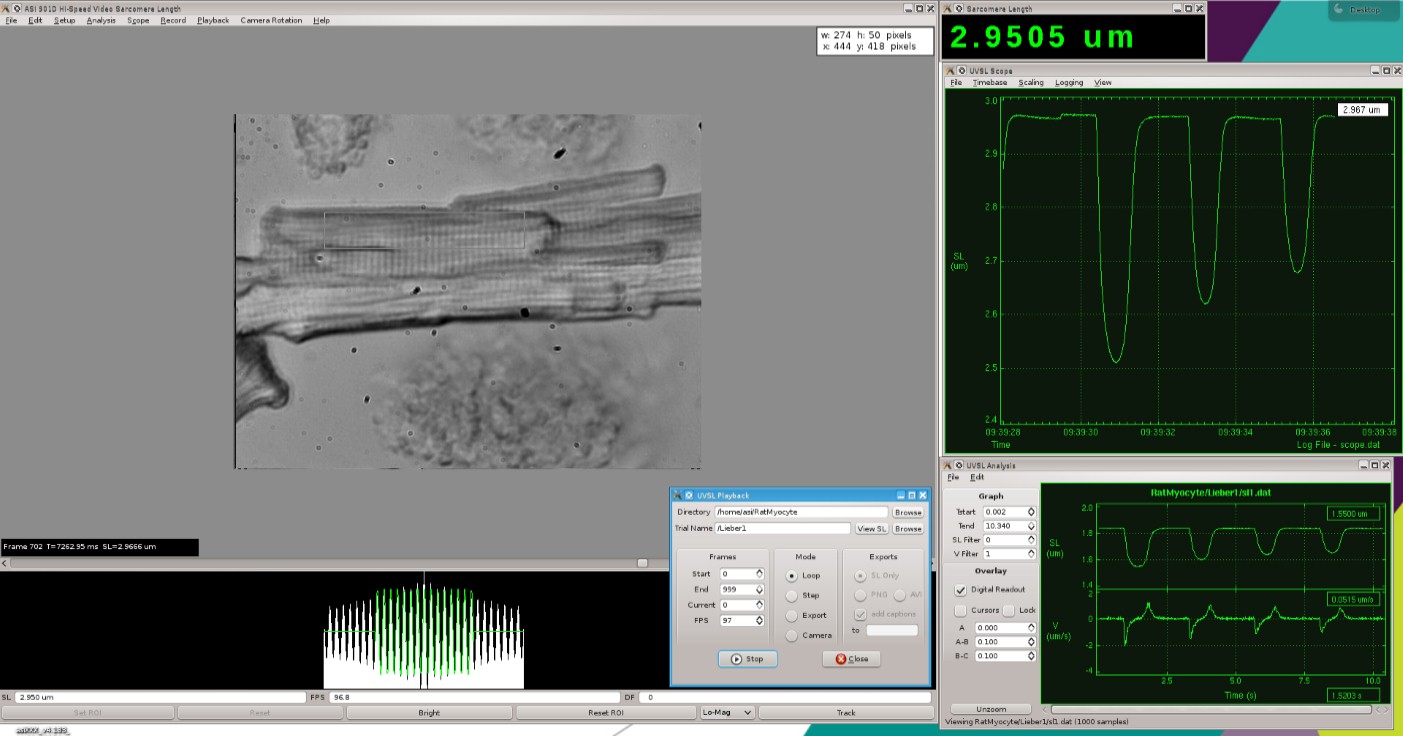
901D: High-Speed Video Sarcomere Length
A simple, turn-key system for measuring sarcomere spacing in isolated muscle preparations.
Learn MoreSpecifications
| Bath | 803B |
|---|---|
| Number of Baths | 8 |
| Bath Volume [µL] | 94 |
| Bath Dimensions [mm] | 6.35 OD x 2.95 D |
| Centerline Distance, X-axis [mm] | 6.35 |
| Centerline Distance, Y-axis [mm] | 6.35 |
| Bottom Cover Slip Dimensions [mm] | 22 W x 40 L x 0.17 T (1 ½) |
| Mounting Slide Dimensions [mm] | 76.2 L x 25.4 W x 1 T |
| Thermoelectric Coolers | 803B |
|---|---|
| Number of TECs | 2 |
| Control Temperature [°C] | 0 – 40 |
| Power [W] | 16 |
| Voltage [V] | 8 |
| Thermocouple Microprobe | 803B |
|---|---|
| Model | Physitemp IT-18 |
| Type | T Thermocouple Copper-Constantan |
| Time Constant [s] | 0.1 |
| Construction | 0.064 mm diameter Teflon tube with sealed tip |
| General Specifications | 803B |
|---|---|
| Weight [gm] | 150 |
| Dimensions [cm] | 11 W x 16 L x 4.8 H |







