Research into the neuromuscular dysfunction and therapeutic options available to patients suffering from muscle wasting or loss of muscle function diseases is ongoing. Thanks to Aurora Scientific equipment, numerous groups have been able to further expand our understanding of these pathologies and how we can improve muscle function. This publication review highlights some of the latest pathophysiological research that utilize our line of equipment.
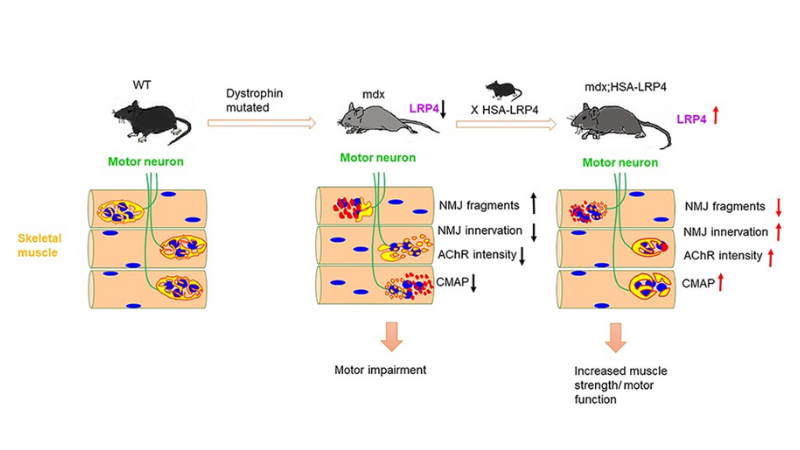
Featured image (©Hui et al. (2021), licensed under CC BY), shows the cross between DMD mice and transgenic LRP4 mice which leads to improved neuromuscular junction function and formation.
Metformin Increases Sarcolemma Integrity And Ameliorates Neuromuscular Deficits In A Murine Model Of Duchenne Muscular Dystrophy
Loss-of-function mutations in the gene that encodes dystrophin disrupts the integrity of muscle fibers that leads to weakness and wasting. As a regulator of energy metabolism, when AMP-activated protein kinase (AMPK) is stimulated, it leads to muscle regeneration and improved function. Therefore, it may be a useful method of inducing therapeutic effects on Duchenne muscular dystrophy (DMD). Metformin is an anti-hyperglycemic drug known to have indirect effects on AMPK. Thus, Dong et al. (2021) sought to determine if metformin could improve muscle function through AMPK in a mouse model.
The authors used an mdx murine mouse model of DMD in their study. By treating the mice with metformin, they were able to study the indirect effects of AMPK stimulation. The authors used our 1300A 3-in-1 Whole Animal System to perform in-vivo twitch and tetanic force measurements on the dorsiflexor group mainly comprised of the Tibialis Anterior (TA). They found that there was an increase in neuromuscular transmission in the mice. Further analysis of mRNA and protein showed that AMPK was upregulated and dystrophin levels were also higher. This indicated that metformin could be a potential therapeutic drug to improve muscle function in DMD patients.
Neuromuscular Junction Transmission Failure Is A Late Phenotype In Aging Mice
Sarcopenia is the loss of muscle function and mass that is abnormally accelerated in older adults. Neurodegeneration is considered to be a factor in sarcopenia. The neuromuscular junction is the bridge between the nervous system and skeletal muscle, and as such, much attention has been focused on the junction as it pertains to aging and muscle function. The authors of this article, Chugh et al. (2020), sought to determine the reliability and fidelity of the neuromuscular junction as age increases. They wanted to narrow down the age-related alterations of the neuromuscular junction; does the dysfunction occur through a direct or indirect process?
The authors investigated the neuromuscular junction across the mouse lifespan by using mice of different ages. The authors used our 1300A 3-in-1 Whole Animal System to investigate in-vivo plantar flexion torque measurements. These contractility recordings showed that mice aged 27 months (the second highest age group in the study, followed by 29 months) demonstrated reduced muscle fiber excitability. Recordings of the neuromuscular junction at that age also showed a reduction. The authors concluded that reduced muscle excitability as a result of neuromuscular junction attenuation could be a target for future therapies to improve muscle function in older adults.
Increasing LRP4 Diminishes Neuromuscular Deficits In A Mouse Model of Duchenne Muscular Dystrophy
In this article the authors, Hui et al. (2021), explored the links between DMD and the neuromuscular junction. The junction is critical for muscular control, and therefore it is not surprising that in DMD patients, there is greater neuromuscular junction dysfunction. The mechanisms behind this decline in function in DMD are not clear, but the effects of reduced neuromuscular junction in DMD are well-documented. Thus, the authors of this article looked at the signaling pathways that are critical in neuromuscular junction formation and maintenance. The protein LRP4 which is a receptor for agrin (a signaling protein that induces junction formation) was reduced in DMD, so the authors hypothesized that LRP4 could be a beneficial protein for DMD.
The authors used our 1300A 3-in-1 Whole Animal System to characterize muscle function in anesthetized mice. The mice the authors used were LRP4 transgenic mice crossed with mdx mice. Muscle twitch and tetanic force was collected, and the authors found that with improved LRP4 expression, the attenuated function and formation of the neuromuscular junction seen in mdx mice was mitigated. This indicates that LRP4, together with agrin signaling, could be another strategy for improving conditions for DMD patients by protecting the function and structure of the neuromuscular junction indirectly.