Aurora Scientific offers several different test systems to evaluate the various structural levels of muscle. However, there are many examples of these systems being used for other types of compliant tissues and materials. Most of these systems are able to work with a multitude of different samples and perform a wide array of experiments.
This blog is meant to be a quick, comprehensive guide to what research has been done with the 1600A – Permeabilized Myocyte System and a primer for anyone searching for the most appropriate muscle test system based on their specific research objectives.
This blog is part of a collection – click below to view other guides by system type:
1200A/1205A Isolated Muscle Systems for Mice and Rats
1300A/1305A/1310A Whole Animals Systems for Mice, Rats, Dogs, Pigs and Other Large Animals
1400A/1405A/1410A/1415A Permeabilized Fiber System
1500A/1510A/1530A Small Intact Isolated Muscle System
1600A – Permeabilized Myocyte System
The 1600A system is very similar to the 1400A in terms of the types of experiments which it can perform. The big differences between the two systems are the type of samples which they are meant to work with. The 1600A is intended to work with only permeabilized cardiac myocytes only. These have some advantages as the myocytes may be cultured, ensuring an abundant supply. A calcium gradient is achieved by creating small wells of activating solution and is manually controlled in this system. Although technically possible, it is not common to use larger samples (such as skeletal fibers) with this system as the automation of the 1400A is a more appealing and higher throughput means of testing larger pieces of de-membranated tissue.
To download a chart of the information below, click here!
Common Samples:
Cardiac Muscle:
- Single de-membranated cardiomyocytes
Common Experiments:
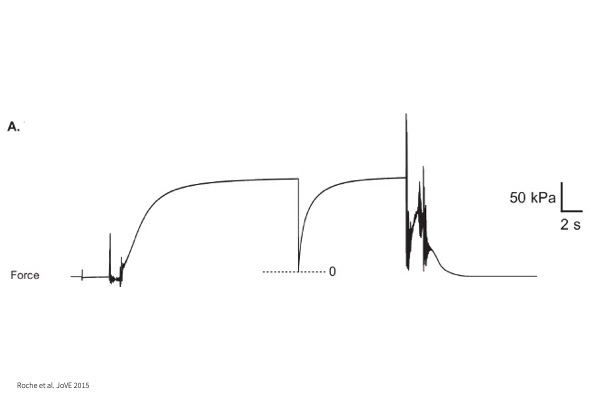
Max Tension:
Tension produced by myocyte when maximally activated with Calcium.
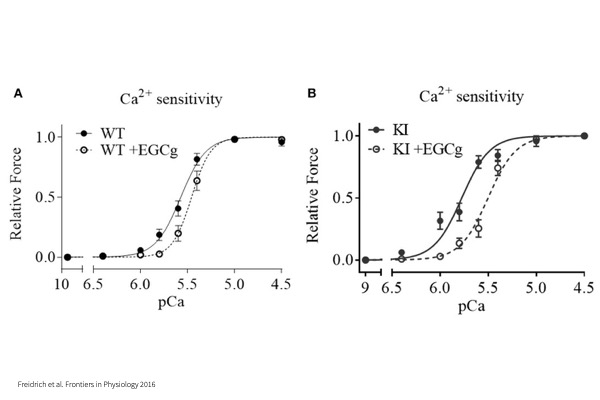
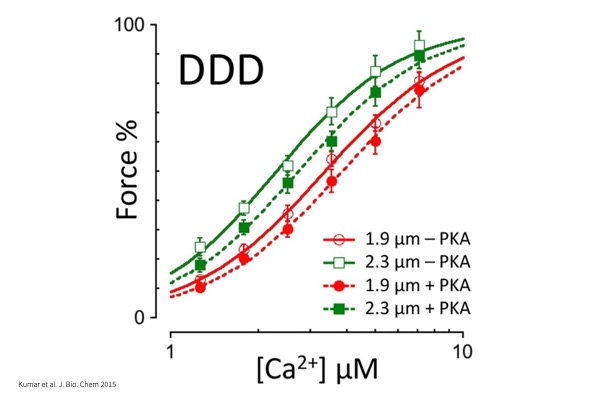
Force – pCa:
Maximum tension produced by myocyte when activated with a series of Calcium concentrations to investigate Calcium sensitivity.
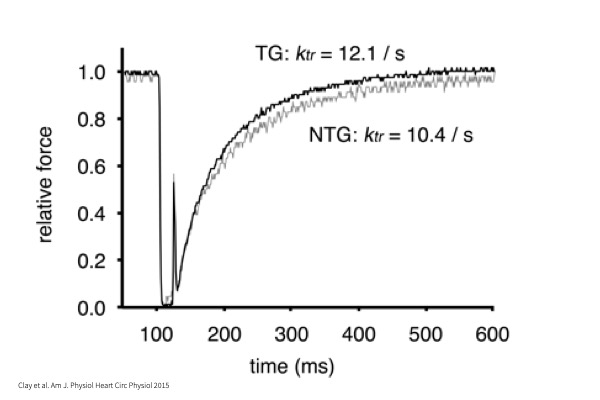
kTR:
Time to tension redevelopment whereby myocyte undergoes maximal activation in Calcium followed by a rapid shortening to zero load and return to original cell length.
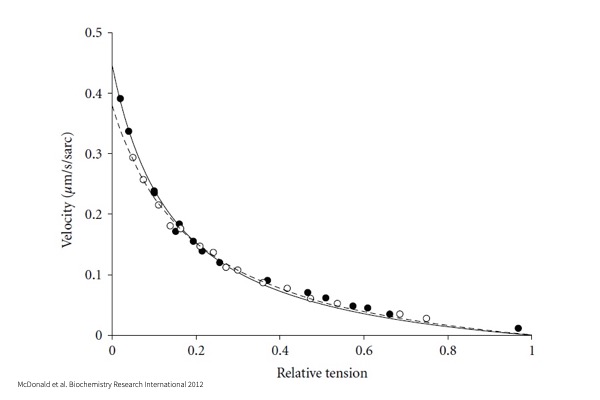
Force-Velocity:
Maximal myocyte activation in Calcium followed by a series of force clamps to a percentage of maximum force, allowing for measurement of shortening velocity.
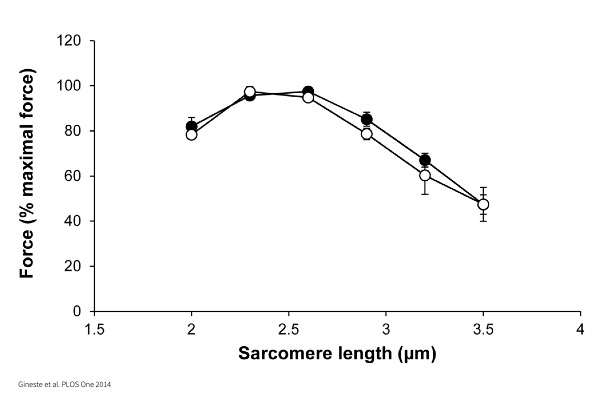
Length – Tension:
Incrementally increasing cell length or sarcomere length to measure maximal force generation.
Frank-Starling (Cardiac):
Length-dependent activation of single myocytes and its effect on Calcium sensitivity.
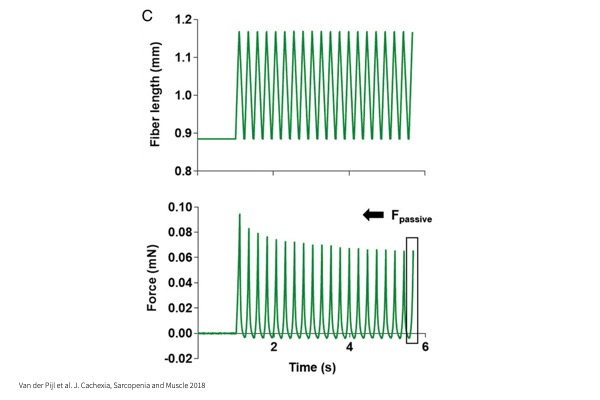
Stiffness:
Passive sinusoidal lengthening and shortening of myocyte to assess the inherent stiffness of the cell.