Aurora Scientific offers several different test systems to evaluate the various structural levels of muscle. However, there are many examples of these systems being used for other types of compliant tissues and materials. Most of these systems are able to work with a multitude of different samples and perform a wide array of experiments.
This blog is meant to be a quick, comprehensive guide to what research has been done with the 1300A/1305A/1310A Whole Animal Systems and a primer for anyone searching for the most appropriate muscle test system based on their specific research objectives.
This blog is part of a collection – click below to view other guides by system type:
1200A/1205A Isolated Muscle Systems for Mice and Rats
1400A/1405A/1410A/1415A Permeabilized Fiber System
1500A/1510A/1530A Small Intact Isolated Muscle System
1600A – Permeabilized Myocyte System
1300A/1305A/1310A Whole Animals Systems for Mice, Rats, Dogs, Pigs and Other Large Animals
Our 1300A system was originally designed solely for performing footplate (in-vivo) experiments in mice and rats. It quickly expanded into a system which could also be used for in-situ experiments and a horizontal in-vitro add on bath was also built. Various versions of the system were later built to accommodate larger animal models up to the size of pigs and dogs. The typical experiments which can be performed are similar to those of the 1200A system family, except that a much broader selection of muscles can be tested by the in-situ method. Those muscles that do not have a surgically accessible tendon or are very large which would have been impossible to test in-vitro are now potentially in play using the in-situ method.
To download a chart of the information below, click here!
Common Samples:
Skeletal Muscle – in-vitro:
- Extensor Digitorum Longus (EDL)
- Soleus
- Plantaris
- Diaphragm
- Lumbricalis
Skeletal Muscle – in-situ:
- Tibialis Anterior (TA)
- Gastrocnemium
- Quadriceps
- Hamstring
- Tongue
- Rotator Cuff
Skeletal Muscle – in-vivo:
- Dorsiflexors (TA, EDL)
- Plantarflexors (Gastroc, Soleus, Plantaris)
- Jaw
- Forefinger (Flexor Digitorum Brevis; FDB)
Artificial Muscle and Connective Tissue:
- Cartilage
- Epithelial
- Tendon
Common Experiments:
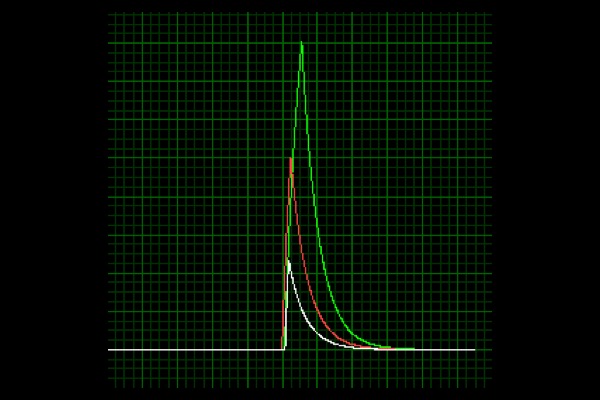
Twitch:
Single impulse designed to elicit contraction of a single or small number of muscle fibers.
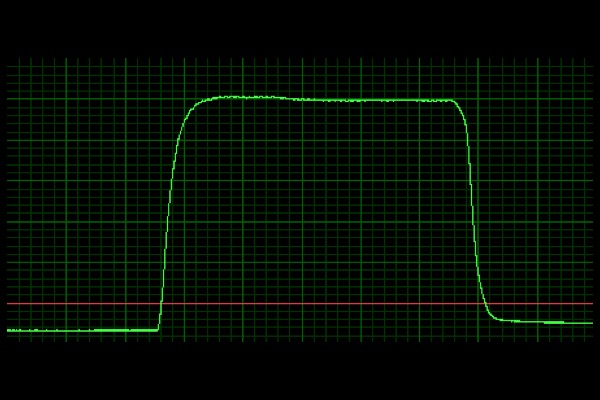
Tetanus:
Multiple electrical impulses in quick succession resulting in temporal summation and complete muscle contraction.
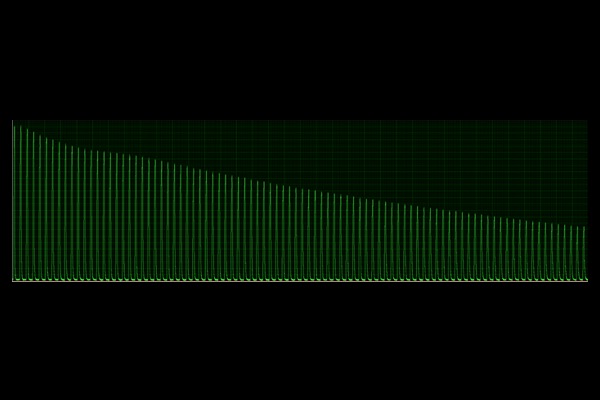
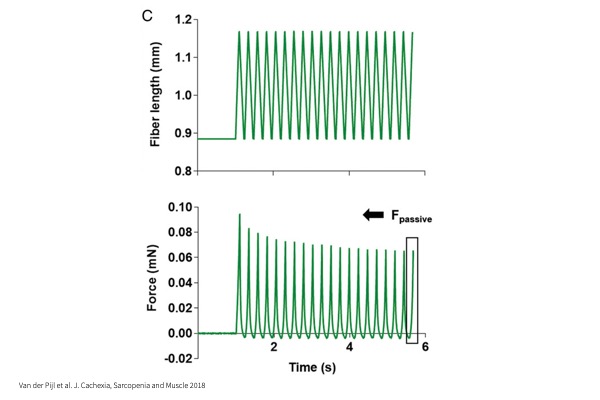
Fatigue:
Sub-maximal tetanic contraction repeated frequently to induce fatigue of the muscle.
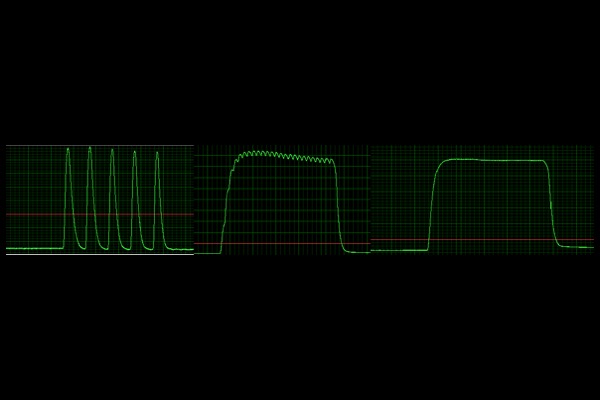
Force Frequency:
Varying rates of stimulation frequencies to assess optimal frequency that elicits maximum tetanic force.
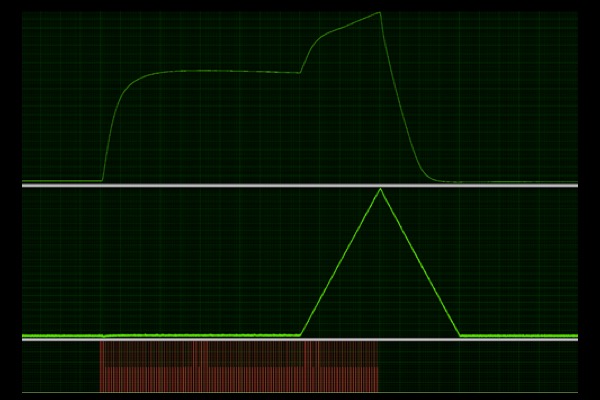
Eccentric:
Active lengthening of muscle during isometric tetanic contraction to induce injury and assess resistance to injury or recovery from it.
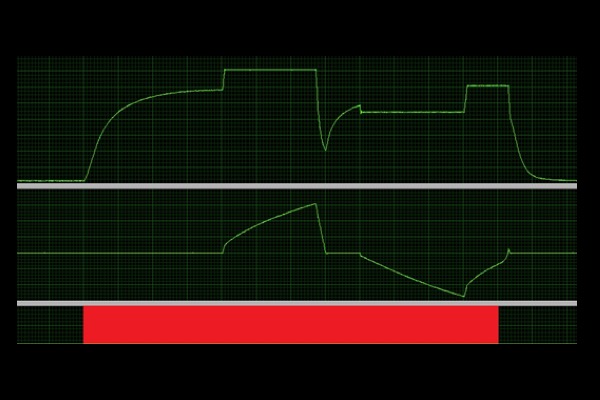
Gait Analysis/Modelling Exercise (Isotonic, Concentric):
Controlling muscle force output (isotonic) to assess shortening velocity of muscle.
Stiffness:
Passive sinusoidal lengthening and shortening of muscle to assess the inherent stiffness of the tissue.
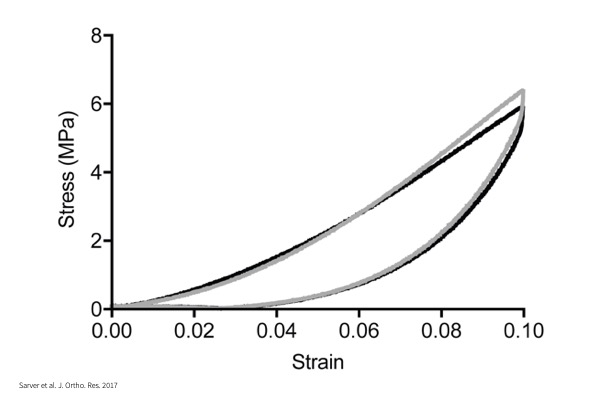
Stress-Strain:
Incremental lengthening of tissue to calculate Young’s modulus of materials.