The 1700A single myofibril system (sometimes referred to as the ‘Penguin’) is commonly used to investigate the active and passive properties of single myofibrils. This system allows researchers to study the thick and thin filament in the purest system possible. However, the myofibrils are tiny and depending on the type of myofibrils tested will produce contractile forces that are a fraction of a micro newton. Any small idiosyncrasies can cause a large artifact when measuring contractile fore. This blog is meant to provide some helpful information to improve the quality of your measurements and increase experimental throughput.
Select the right glue for the job
Attaching myofibrils is very similar to attaching permeabilized myocytes and the adhesives used for myofibrils are similar, or in many cases identical, to those used for myocytes. Please see our tips and tricks blog for the 1600A system for more information about these adhesives.
However, it is possible to attach these myofibrils without the use of any adhesive at all simply through electrostatic interaction between tissue and glass microneedles. This typically requires that the tooling be polished for best adhesion properties but offers an alternative to cleaning the tooling after each application with glue. Sharpened microneedles have been used to pierce the myofibril, but this is not possible for both ends of the system as the AFM cantilever cannot be physically sharpened.
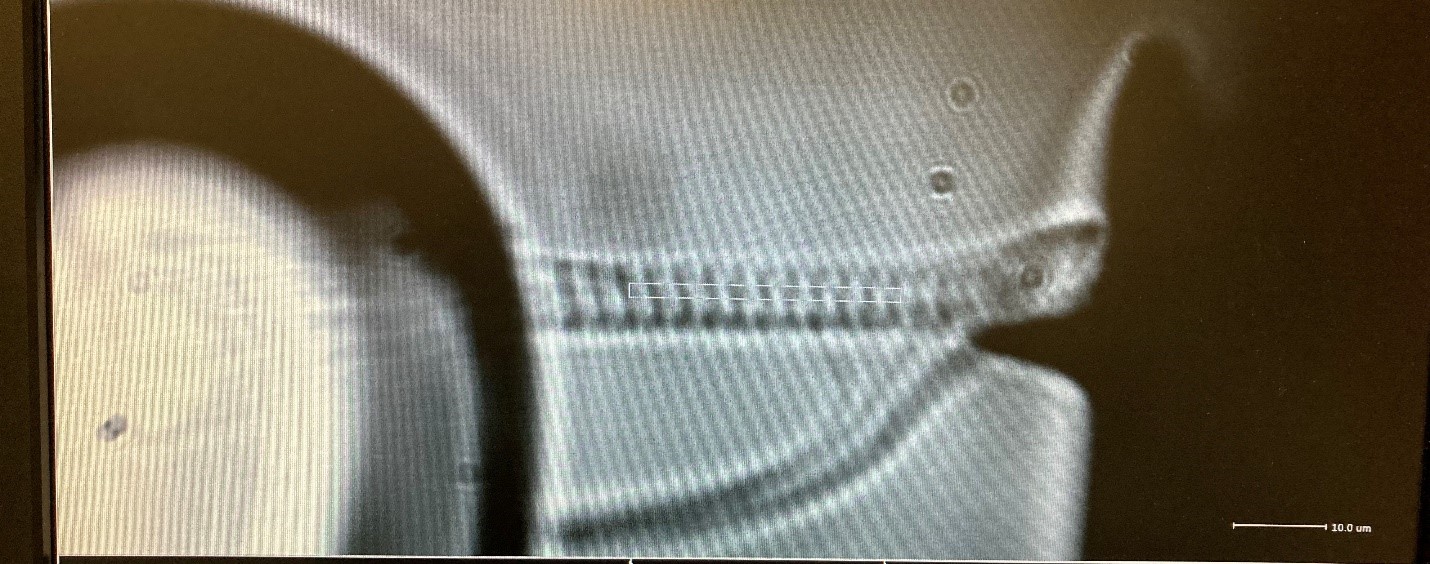
Figure 1: Myofibril attached without glue to AFM and piezo motor probe. Photo courtesy of Harris Lab University of Arizona.
Optimizing Calibration sets you up for success
Force measurements made with the system are based on measurement of deflected light of a conjugated laser which reflects off a polished AFM chip. Thus, if the laser is focused on different spots on the AFM chip, the amount of deflected light will differ and a consistent measure of force between two different myofibrils will not be possible. There are several techniques that will help the user optimize not only the magnitude of the signal but also the repeatability.
- Make use of the 820A XYZ motion controller
Many researchers who have experience working with tooling attached to motorized micropositioners are used to making manual adjustments of position either via joystick or keypad. Those with sufficient experience can adeptly navigate the probes/tooling quickly and accurately use them to attach to a sample. However, placing the tooling back in the exact calibration spot in 3D space after navigating to pick up a myofibril requires a level of expertise that few can achieve manually. The 820A XYZ motion controller provides a means of remembering various points in 3D space which the user can return to at their convenience with a precision of less than 1um.
Additionally, the motion controller has a software interface that is fully integrated with the data acquisition software. It allows movements of very precise magnitudes to be made which is useful for fine adjustments and delicate operations such as sample attachment.
- Location, Location, Location
Since we have the ability to make precise movements of the micropositioners, we should strive to optimize the relative position of the AFM cantilever in 3D space. The focal point of the conjugated laser spot is set to be approximately 500-600um from the face of the optical periscope. To set this distance we should focus on the bottom edge of the optical periscope and use a lower powered objective to set the cantilever distance from it. This distance can be easily measured with the included HSVL software. The size of individual myofibrils typically requires a very high powered objective to be used in order to visualize clearly, which typically means a small FOV. It can help to have the periscope in the FOV for reference, but this may require adjusting the focal distance of the laser inside the 470A sensor head. Please speak with Aurora Scientific directly about customizing this distance based on the needs of your existing microscope.

Figure 2: Optical Periscope and AFM tooling shown together through microscope eyepieces.
Use the fit and modelling tools in the 600A calibration app to your advantage
To obtain a stiffness value for the AFM cantilever we must physically bend it and measure the change in signal. This is best accomplished by positioning the probe tip of the 340A piezo motor in contact with the AFM. As the AFM is delicate and it is difficult to determine the exact point of good contact between the two probes, it may be advantageous to experiment with the number of steps the piezo motor will take and their individual size. You may also encounter sudden portions of non-linearity in the signal with respect to cantilever deflection. This is generally due to lack of contact between the piezo motor probe and the AFM. However, to salvage the calibration these points may be otherwise left out of the linear fit. (See figure 3 & 4).
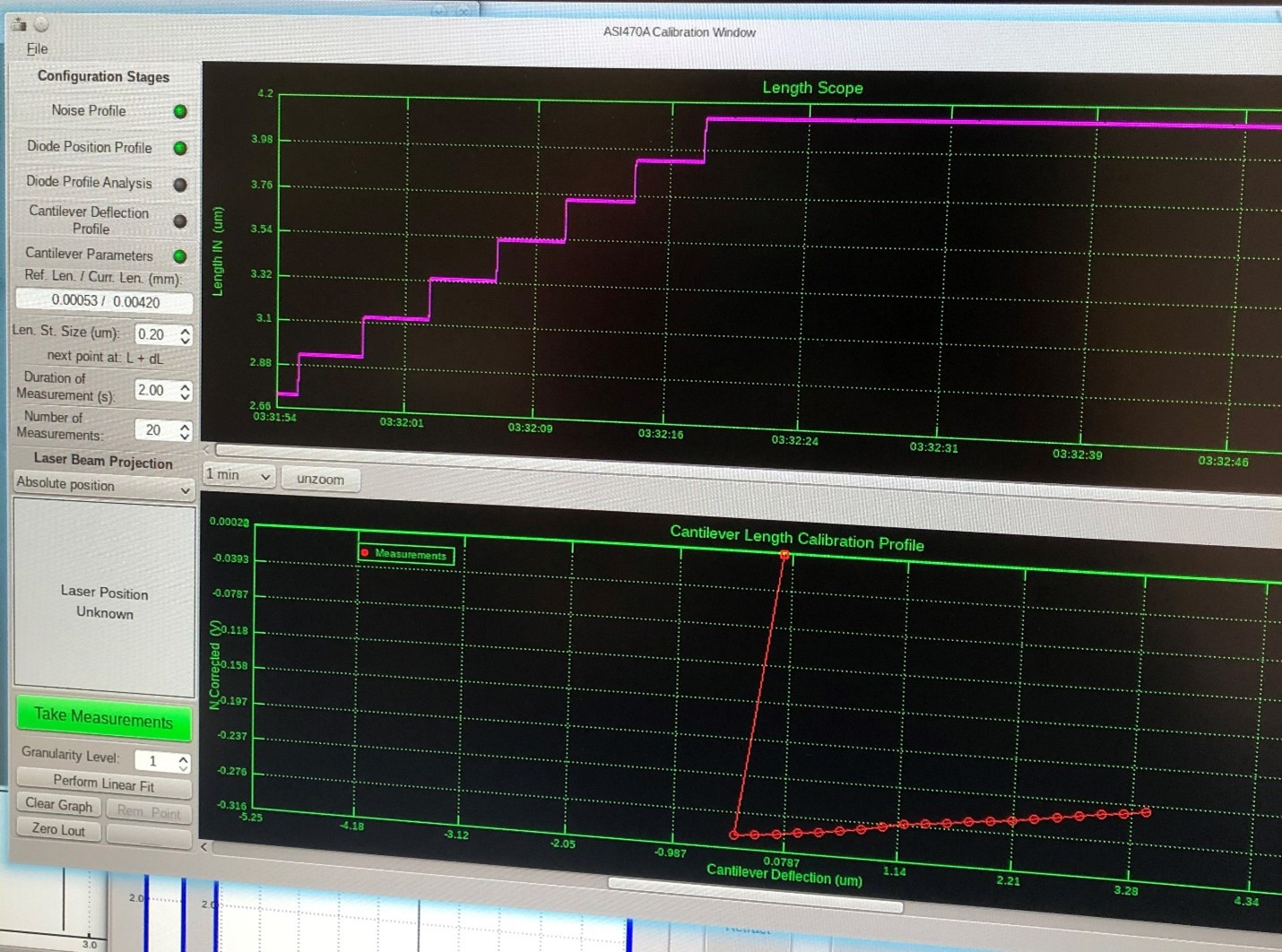
Figure 3: Non-linearity in stepwise calibration.
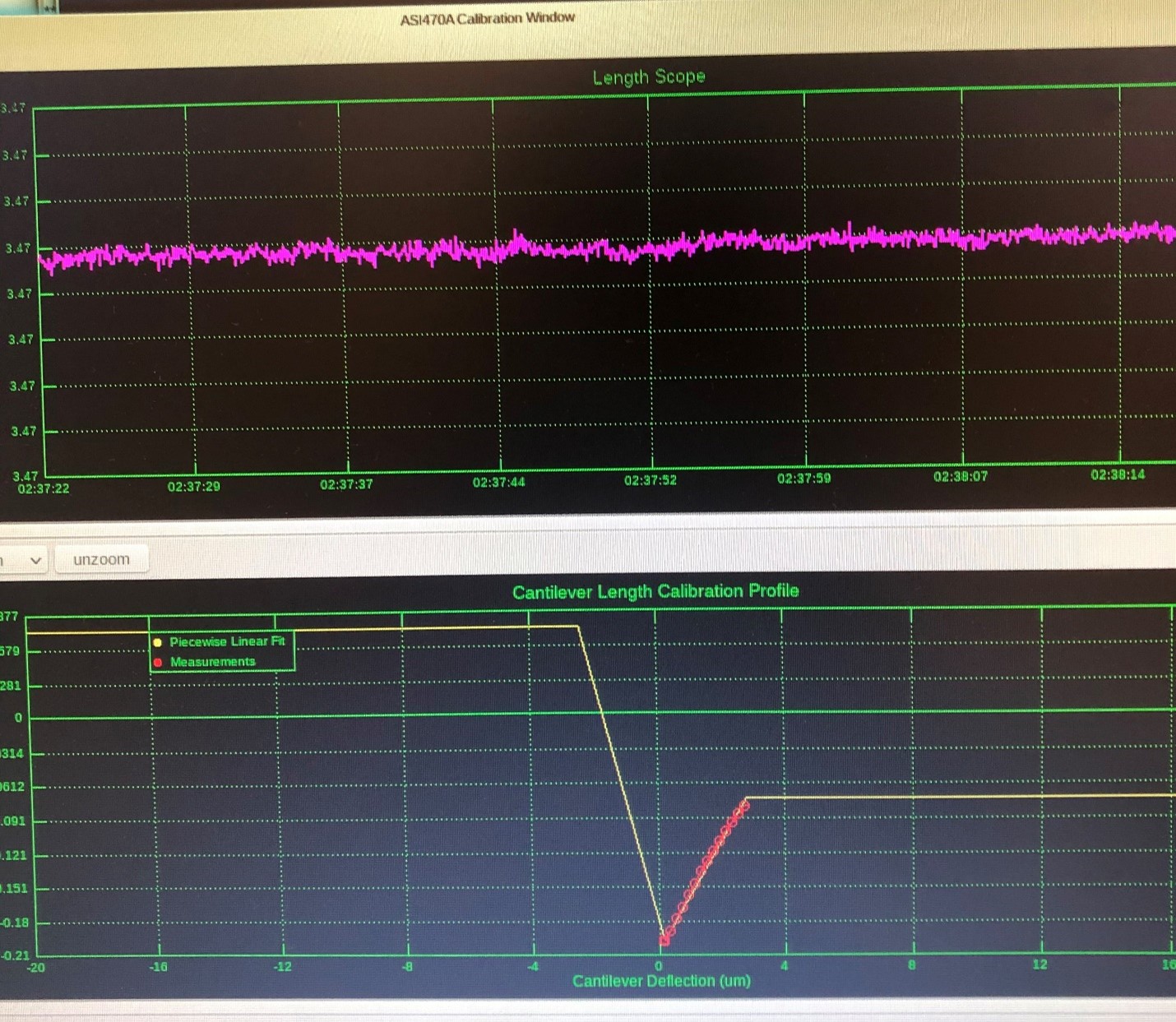
Figure 4: Non-linearities removed with calibration editor.
Use the Live Monitors to make fine adjustments in signal magnitude
We have already seen how the 820A XYZ positioner coupled with the visual feedback of the HVSL camera can greatly assist in facilitating precise, relative positioning. However, fine adjustments of reflected light can be tuned by monitoring the Sum signal in the live monitor while making fine adjustments of position. For instance, we can move the AFM in small steps towards and away from the optical periscope and monitor the Sum signal to find the point at which it peaks. Increments of 5-10um can make a reasonable difference to signal magnitude.
Additionally, it can be difficult to know the exact position of the focused laser spot on the AFM chip itself. Since the diameter of the laser spot (even at its perfect focal point) is still relatively large compared to the width and overall size of the AFM chip, small movements of the AFM side to side and up and down are the only way to determine if the center of the spot is fully on the flexing arm of the AFM itself. Partially focusing on the spot on the arm and body together, or having the laser spot off center from the AFM will still yield a change in signal, but its magnitude may be far below optimal and an adjustment of tens of microns with the assistance of the live monitor may boost the signal significantly.
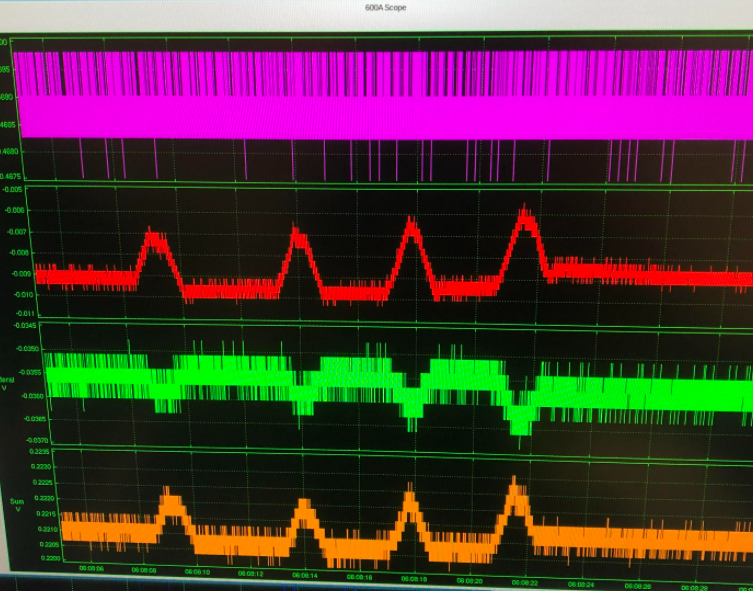
Figure 5: Incremental movements of a few microns affecting the magnitude of the signal.
Stay tuned to this blog for more upcoming tips and tricks on a wide variety of products and topics!