As always, the entire team at Aurora Scientific has enjoyed reading and learning this year about the fantastic research and novel discoveries that are being made by scientists that incorporate our products into their work. While we celebrate all published work by our customers, here are 7 publications selected by our staff that really stood out in 2019!
We would also like to take this opportunity to say thank you and offer congratulations to all of our customers who work tirelessly on their research. If not for you, we would not have the opportunity to innovate and play a role in the research and discovery process . We look forward to reading all of your latest and greatest publications in the new year!
Continue scrolling, or select from the list to jump to a publication of interest.
- Elastic tissue forces mask muscle fiber forces underlying muscle spindle Ia afferent firing rates in stretch of relaxed rat muscle
- Deleting Full Length Titin Versus the Titin M-Band Region Leads to Differential Mechanosignaling and Cardiac Phenotypes
- Development of Novel Micro-dystrophins with Enhanced Functionality
- Impaired skeletal muscle mitochondrial pyruvate uptake rewires glucose metabolism to drive whole-body leanness
- Reduced mitochondrial lipid oxidation leads to fat accumulation in myosteatosis
- Encoding of wind direction by central neurons in Drosophila
- Fibro-Adipogenic Remodeling of the Diaphragm in Obesity-Associated Respiratory Dysfunction
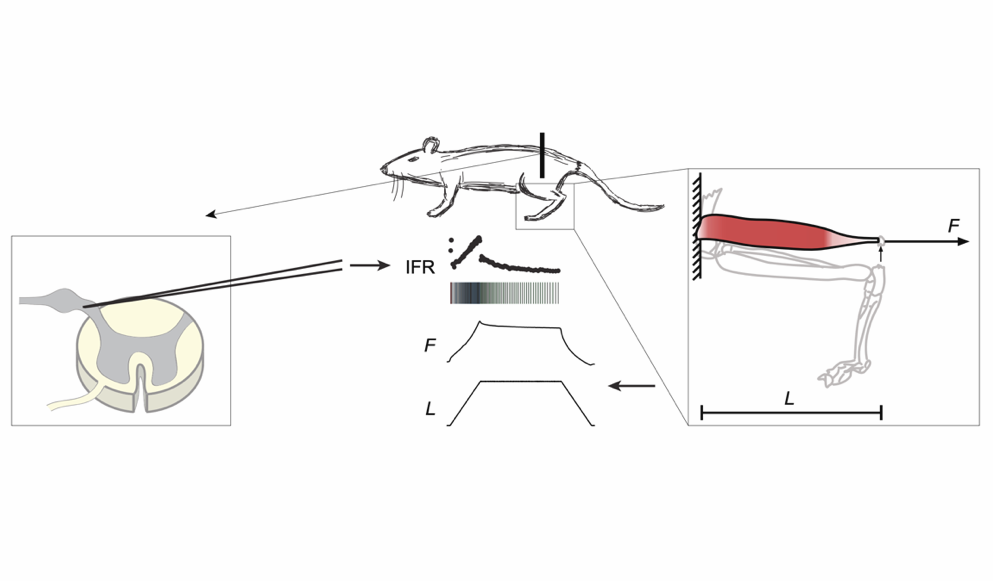
Elastic tissue forces mask muscle fiber forces underlying muscle spindle Ia afferent firing rates in stretch of relaxed rat muscle
In this study, the authors hypothesized that in rats, the firing rate of muscle spindles during stretch of relaxed muscle mirrors muscle fiber forces, but that these forces are masked by the elastic force of extracellular tissue.
To investigate this, the researchers, Blum, Nardelli, Cope and Ting, measured muscle force in response to stretch in vivo in rats. They controlled and measured muscle length and resultant force using the Aurora Scientific 305C-LR dual-mode lever. The force contribution of the extracellular tissue was estimated using an exponentially elastic tissue model, and the muscle fiber force was estimated by analytically removing ECM force from the measured force.
They found that the resulting muscle forces were consistent with thixotropy, with the muscle exhibiting greater force in the first stretch compared with subsequent stretches. This is significant, as previous studies (which did not account for the contributions of ECM) had found that muscle forces could not be explained directly by thixotropy.
These results suggest that history-dependent muscle spindle firing and muscle force in rats arises from forces owing to thixotropy in muscle fibers.
Deleting Full Length Titin Versus the Titin M-Band Region Leads to Differential Mechanosignaling and Cardiac Phenotypes
In this study the authors, Radke et. al. investigated the functional differences between mice with titin deficiencies vs. dysfunction. To do this, they used the Cre/lox recombination system to generate two knockouts – one to model titin deficiency (knockout of exon 2) and one to model titin dysfunction (knockout of an M-band exon that is critical for sarcomeric integration).
To assess the effects of the knockouts, they evaluated cardiomyocyte mechanics. Isolated cardiomyocytes were secured between the Aurora Scientific 406A force transducer and 315C-I high-speed length controller. Data were also collected using video sarcomere length (900B VSL) and 600A data acquisition and analysis system, respectively.
Additionally, the authors used echocardiography to assess intact cardiac function, histology and electron microscopy to visualize the tissue structure, and real-time PCR for expressional analysis of the titin isoforms.
Unsurprisingly, deficiency and dysfunction of titin both led to sarcomere disassembly, reduced strength and early lethality. However, they found that, in mice with dysfunctional titin, cardiomyocytes maintained their elastic properties, while passive stiffness was reduced in mice with reduced levels of titin. Cardiac phenotypes were also significantly different between the knockout groups: loss of titin led to dilated cardiomyopathy with combined systolic and diastolic dysfunction, while titin dysfunction led to cardiac atrophy and preserved function.
In humans, mutations of the titin gene represents the most common genetic basis of heart disease. As we gain a better understanding of the phenotypic differences between different forms of titin, we can improve diagnostics, clinical decision-making and therapies for affected individuals.
Development of Novel Micro-dystrophins with Enhanced Functionality
Duchenne muscular dystrophy (DMD) results from aberrant or absent expression of dystrophin. As a currently incurable recessive genetic disorder, it is a candidate for gene therapy. However, the dystrophin gene is quite large, and the carrying capacity of adeno-associated viral (AAV) vectors is relatively small.
In this study, Ramos et. al. describe the development and evaluation of several micro-dystrophins (µDys), versions of the protein that have been sufficiently reduced in size in order to be carried by AAV. The research team’s previous designs of µDys displayed significant (though still incomplete) function in striated muscle.
To assess the effectiveness of the proteins, vectors were delivered to dystrophic mdx4cv mice via intramuscular injection. To assess muscle force generation and susceptibility to injury, they subjected muscle to a series of progressively increasing length changes under stimulation using the Aurora Scientific 305C-LR dual-mode lever system and 701C electrical stimulator respectively.
The study was a great success: two of their µDys constructs led to increased force generation compared with older designs. Excitingly, one of them is currently being evaluated in a DMD clinical trial!
Impaired skeletal muscle mitochondrial pyruvate uptake rewires glucose metabolism to drive whole-body leanness
In this study, the authors investigated the effects of decreasing muscle mitochondrial pyruvate uptake on muscle and systemic glucose metabolism. To do this, they generated mitochondrial pyruvate carrier (MPC) knockout mice. Under normal conditions, MPC mediates the transport of pyruvate into mitochondria, linking glycolysis with oxidative phosphorylation.
In these knockout mice, they used a series of assays at the molecular and tissue-level. At the whole-muscle level, force production of the lower hind limb was measured using the Aurora Scientific 305C dual-mode lever.
They found that, surprisingly, MPC disruption did not hinder glucose uptake in muscle tissue. Instead, as the authors note,
“it evoked complex, interconnected changes in muscle and systemic metabolism leading to increased whole-body insulin sensitivity, increased muscle glucose uptake, and attenuation of obesity and T2D.”
Knockout mice had greatly reduced adipose tissue with no loss of muscle mass and, notably, strength retention. Importantly, even on a normal diet, deletion of MPC led to an acceleration of fat loss in obese mice.
These data suggest that MPC may represent a possible target for therapies to treat obesity and type 2 diabetes. However, the authors do note that much more work must be done to better understand how modulation of MPC affects the function of tissues like the brain, heart and kidney.
Reduced mitochondrial lipid oxidation leads to fat accumulation in myosteatosis
In this study, Gumucio et. al. set out to investigate pathological changes resulting in lipid accumulation in injured muscle fibers. To this end, they used a rat model of rotator cuff injury. Muscle samples were collected 10, 30 or 60 days after the injury, and tissue was analyzed using a variety of assays including RNA sequencing, metabolomics, lipidomics and proteomics to identify the possible mechanisms and processes underlying the dysregulation following muscle tear. In addition, muscle fiber contractility was measured using an Aurora Scientific permeabilized fiber system.
They identified that mitochondrial function was likely disrupted after injury. Given the mitochondria’s role in lipid metabolism, they performed biochemical and imaging studies and determined that mitochondrial dysfunction in particular likely leads to the accumulation of lipid in myosteatosis.
These results are relevant for the clinical treatment of muscle injury. The authors note that our ability to treat mitochondrial dysfunction and lipid oxidation is limited, so improvements in normalizing mitochondrial function in injured tissue is an avenue to potentially improve clinical outcomes for rotator cuff injury and other myosteatosis-related conditions.
Encoding of wind direction by central neurons in Drosophila
In this study, the authors, a group from New York University Neuroscience Institute, investigated neural basis for orientation using wind direction in Drosophila. Wind direction is an especially important sensory cue for flies, as wind direction often provides a better indication of an odor’s source compared to odor concentration. And, of course, a fly’s ability to accurately follow its ‘nose’ means the difference between finding its next meal or not.
To determine how antennae work together to do help flies orient themselves in wind, the researchers put Drosophila in a makeshift wind tunnel and manipulated antennae in various ways. In behavioral experiments, they blocked the mechanosensory input from the antennae by pinching them off. After a recovery period, they measured the flies’ angular velocities as they set out to find the source of an odor pulse. To measure the precise amount of odor delivered, they used the Aurora Scientific miniature photo-ionization detector (miniPID). Alongside the tightly controlled behavioral conditions, they used electrophysiological methods to make single-cell recordings from individual neurons involved in olfactory search behavior and orientation to wind.
The authors showed that neural input from both antennae is required to orient themselves upwind during olfactory search behavior. However, they found that one antenna is sufficient for flies to orient themselves partially, and these flies adjust their behavior to compensate. Importantly, the authors identified a new class of neurons which they have called WPNs. These neurons encode wind orientation; they are excited by ipsilateral wind and inhibited by contralateral wind.
These results highlight the importance of wind orientation for Drosophila and help define the neural pathways used to translate mechanosensor information into behavior.
Fibro-Adipogenic Remodelling of the Diaphragm in Obesity-Associated Respiratory Dysfunction
It is well known that in obesity, respiratory dysfunction is a common complication. While this is largely due to increased adipose mass physically restricting expansion of the lungs, the effect that obesity has on the diaphragm is not as well-characterized.
In this study, Buras et. al. investigated how obesity affects the diaphragm function in mice. The researchers induced obesity using a high-fat diet for 1, 3 or 6 months, then used a variety of tests to evaluate diaphragm function including ultrasonography as well as assays at the tissue-, cell-, and molecular-level.
To test isometric force, they used an Aurora Scientific 305C dual-mode lever system as well as an Aurora Scientific 701C biphasic current stimulator. Together, contraction and relaxation times, muscle cross-sectional area, and specific force were calculated.
They found that in mice, long-term obesity led to impaired diaphragm function, both in vivo and ex vivo. They also demonstrate a correlation between fibro-adipogenic changes and diaphragm dysfunction: there was a temporal match between impaired muscle function and an increase of adipose tissue, collagen and intradiaphragmatic fibro-adipogenic progenitors.
They conclude that their identification of the temporal correlation between dysfunction and fibro-adipogenic progenitor-mediated remodeling provides a framework for better understanding how obesity can cause respiratory impairment.