February notably marks “Heart Month”, aiming to highlight the importance of cardiovascular health and its critical implications on our overall wellbeing. Tackling the heart of the issue, several cardiovascular studies stand out as key players within the preclinical research landscape. The following publication review covers recent advancements in the field, including a promising therapeutic drug candidate for heart failure as well as novel insights into the cardiovascular biology of Marfan syndrome and muscle contraction regulation.
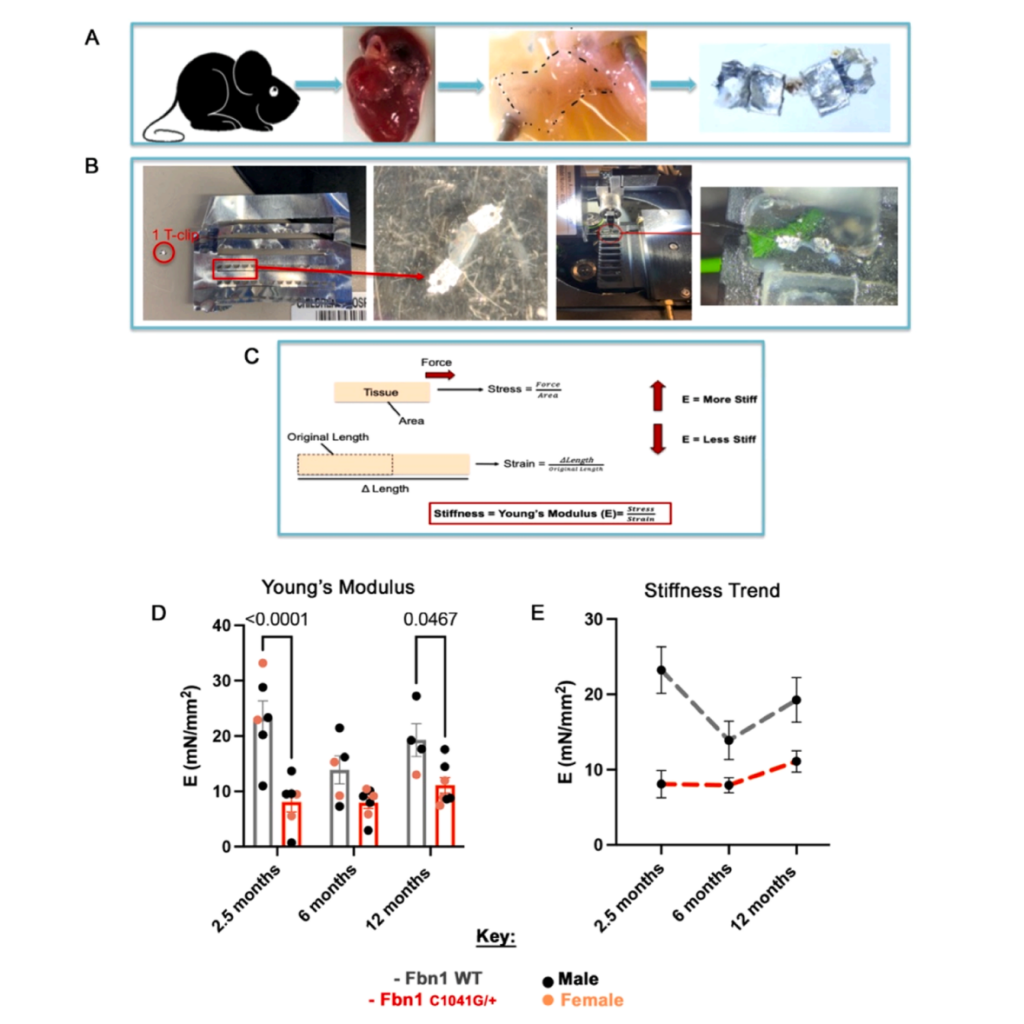
Featured image (Gonzalez et al. (2024), ©2024 Elsevier B.V. All rights reserved.) demonstrating how tissue mechanical testing reveals reduced stiffness of Fbn1C1041G/+ Marfan syndrome mitral valve disease. A) Schematic of the method used for mechanical testing. B) T-clip mounting onto the system. C) Young’s modulus as an indicator of stiffness. D) Stiffness of Fbn1C1041G/+ MFS mitral valve leaflets relative to WT controls. E) Fbn1C1041G/+ MFS mice stiffness over time.
Acute and chronic effects of levosimendan in the ZSF1 obese rat model of heart failure with preserved ejection fraction
The rise of chronic diseases, ageing populations, and resulting demographic shifts have pushed heart failure (HF) conditions to the forefront of cardiovascular disease research. Heart failure with preserved ejection fraction (HFpEF) has been of particular concern, characterized by impaired relaxation and compliance of the heart, cardiovascular stiffness, preserved ejection fraction, and systemic changes including inflammation, endothelial dysfunction, and metabolic abnormalities. Due to this, patients often experience shortness of breath, orthopnea, fatigue, reduced exercise tolerance, and edema, with worsening prognoses over time. Despite the detrimental effects of HFpEF and the alarming rise in its prevalence, treatment options remain strikingly limited.
In an effort to advance the therapeutic landscape of HFpEF, Moreira-Costa et al. (2024) investigated the acute and chronic myocardial effects of an inodilator, termed levosimendan, in a ZSF1 obese rat model of HFpEF. Twenty-week-old male mice were chronically treated with either levosimendan or a vehicle control for 6 weeks, followed by comprehensive phenotypic assessments. At the mechanical level, the group utilized Aurora Scientific’s video sarcomere length imaging software, 900B VSL, and attached skinned left ventricular cardiomyocytes to the 403A Force Transducer and 315C-I High-Speed Length Controller. The 600A Digital Controller was then used to digitally adjust cell length, and the passive and active forces in both relaxing and activating solutions was measured at increasing sarcomere lengths.
Notably, Moreira-Costa et al. (2024) found that skinned myocytes from the treated mice showed a downward shift in sarcomere length-passive tension relationship. In fact, rats chronically treated with levosimendan showed an overall improvement in cardiovascular function, including decreased systemic arterial pressures, raised cardiac index, and enhanced left ventricular relaxation and compliance compared to vehicle-treated controls. As such, these findings provide a promising preclinical framework from which to propose future clinical testing of levosimendan as a potential therapy for HFpEF patients.
Dynamic changes in mitral valve extracellular matrix, tissue mechanics and function in a mouse model of Marfan syndrome
Marfan syndrome (MFS) is a genetic condition that impacts the body’s connective tissue and is caused by mutations in the FBN1 gene. Importantly, more than 50% of MFS patients have an incidence of mitral valve disease (MVD), characterized by structural changes in the heart’s mitral valve. Although progressive MVD has been well documented within the field, the extracellular matrix (ECM) mechanisms underlying the progressive valve degeneration remain critically understudied. As such, Gonzalez et al. (2024) sought to assess the structural, functional and mechanical changes in the mitral valve in MFS mouse models with progressive MVD.
Echocardiography, mechanical assessments, histological analyses, and gene expression experiments were performed to uncover the mechanistic links in MVD progression. To strategically assess the mechanical properties of isolated mouse mitral valve (MV) leaflets, Gonzalez et al. (2024) first measured the specific leaflet lengths and widths before placing them on T-Clips. The prepared samples were then placed onto Aurora Scientific’s 1400A Permeabilized Fiber System, where they underwent a sequential 20% uniaxial stretch. After 3-5 tests, a force versus distance curve was produced for each run using the 600A Digital Controller.
Upon analysis, the group found decreased stiffness of MFS MVs at all stages when compared with controls. Moreover, early prevalence of proteoglycans followed by increased collagen structure and organization were observed as the disease progressed, with ECM mechanics, composition and organization preceding functional abnormalities in MFS MVs. Importantly, these findings provide novel insights into the dynamic regulation of MV structure, tissue mechanics, and function in progressive MVD in MFS mice.
The structural OFF and ON states of myosin can be decoupled from the biochemical super- and disordered-relaxed states
The classical thin filament regulation of muscle contraction, describing the formation of acto-myosin cross bridges has been well documented in the field. However, further investigations have shed light on the central role of both thick and thin filament regulation on muscle contraction modulation. We now know that under relaxed conditions, myosin may reside either in a high-energy-consuming disordered-relaxed (DRX) state available for actin binding and force generation, or in an energy-sparing super-relaxed (SRX) state unavailable for actin binding. In addition, X-ray diffraction studies have shown that many myosin heads are in a quasi-helically ordered OFF state in a resting muscle, and that this helical ordering is lost when myosin heads are turned ON for contraction.
As of late, it has been assumed that myosin heads in SRX and DRX states are equivalent to the OFF and ON states, respectively, and the terms have been used interchangeably across contraction studies. To address this, Jani et al. (2024) investigated the relationship between structurally defined states of myosin (OFF and ON states) and biochemically defined states (DRX and SRX) in muscle contraction regulation. Using the myosin activators, omecamtiv mecarbil (OM) and piperine, in relaxed porcine myocardium, Jani et al. (2024) use X-ray diffraction and ATP turnover assays to track the structural and biochemical transitions of myosin heads. To prepare the muscle for X-ray diffraction, porcine myocardium strips were skinned and the fiber bundles further dissected for attachment to aluminum T-Clips. The muscle was then incubated in a customized chamber, with one end attached to Aurora Scientific’s 402B Force Transducer. Through laser diffraction adjustment, X-ray diffraction was performed at a sarcomere length of 2.3 µm.
Importantly, Jani et al. (2024) found that while OM and piperine shift myosin heads from the OFF to the ON state, there are no significant changes in the population of myosin heads in the SRX and DRX states. This suggests that the biochemically defined SRX and DRX states can be distinct from the structurally defined OFF and ON states, underscoring the critical need to investigate and interpret these transitions separately.
Conclusions
Recent advances in cardiovascular research have pumped out promising therapeutic and mechanistic implications within the field. These studies by Moreira-Costa et al. (2024), Gonzalez et al. (2024), and Jani et al. (2024) ultimately contribute to our understanding of cardiovascular health and the underlying mechanisms at the heart’s core.