Tissue engineering is at the forefront of regenerative medicine, offering immense promise for restoring or enhancing the function of impaired tissues and organs. While tissue constructs remain costly in clinical settings, their use as a research tool holds vast potential for drug discovery, aiding and expediting the screening of drug candidates. In this publication review, we will discuss three recent publications leveraging specialized equipment from Aurora Scientific to advance the innovative field of engineered tissue constructs.
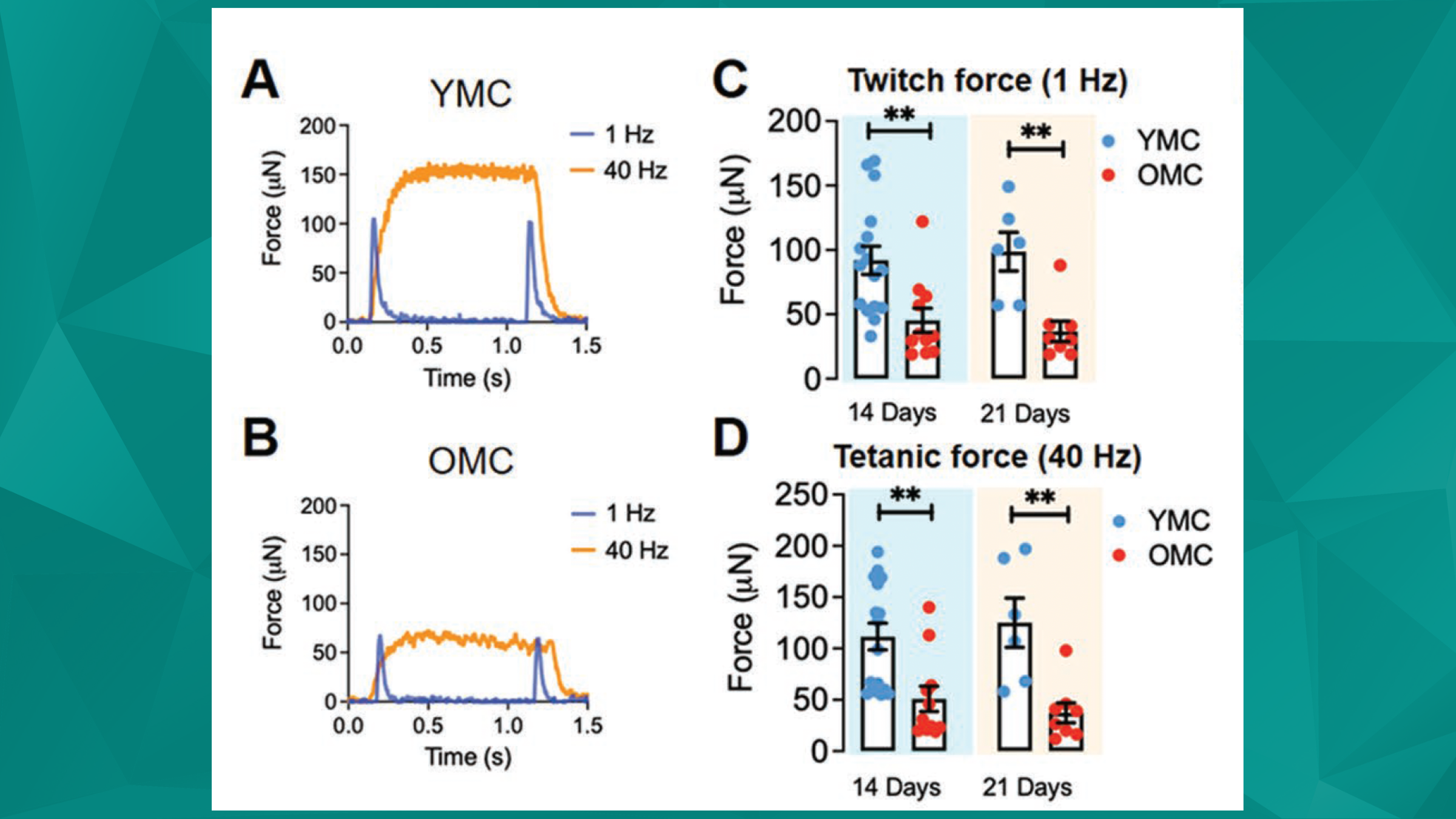
Featured image (©Wang et al. (2023), licensed under CC BY-NC-ND 4.0), showing that old muscle constructs displayed limited contractile function and impaired regenerative capacity. A,B) Representative twitch (1 Hz) and tetanic (40 Hz) force traces of YMC (young muscle constructs) and OMC (old muscle constructs) after 14 days of differentiation. C,D) Average amplitudes of twitch and tetanic forces generated by YMC and OMC after 14 or 21 days of differentiation.
Epicardially Secreted Fibronectin in 3D-Engineered Heart Tissues
Ischemic heart failure, also known as cardiac ischemia or ischemic cardiomyopathy, is the result of reduced blood flow to the heart, typically caused by coronary artery disease. The damage from this condition occurs due to an irreversible loss of cardiomyocytes, however previous preclinical studies have demonstrated that remuscularizing hearts with human pluripotent stem cell-derived cardiomyocytes (hPSC-CM) can restore cardiac function. Other studies show that incorporating epicardial cells into engineered heart tissues improves cardiomyocyte maturation, with the exact mechanism remaining unknown until recently.
Ong et al. (2023) sought to characterize this mechanism by determining the effects of epicardial fibronectin (FN1) as an intermediary between epicardial-cardiomyocyte crosstalk, assessing its role in driving hPSC-derived cardiomyocyte maturation in a 3D engineered heart tissue. Using the 400A Force Transducer and the 312B High-Speed Length Controller, the mechanical properties of the engineered heart tissues were assessed. Loss of FN1 resulted in immature cardiomyocytes with reduced contractile functionality and inefficient calcium handling. These findings thereby demonstrate the importance of FN1 as a regulator of cardiomyocyte maturation in 3D-engineered heart tissues, helping guide future cardiac tissue engineering.
Remodeled 3D eX vivo Muscle Engineered Tissue (X-MET)
Following injury, the adult heart has a limited capacity for self-repair, with cell transplantation and tissue engineering emerging as potential therapeutic options. While several stem cell populations have been used to treat infarcted myocardia, transplanted cells often struggle to establish functional connections with host cardiomyocytes. Consentino et al. (2023) propose a new experimental tool, named 3D eX vivo Muscle Engineered Tissue (X-MET) to help delineate the contribution of particular mechanical stimuli in triggering functional remodeling of the heart tissues.
To demonstrate the mechanical properties of the heart tissue, the 300B Dual-Mode Lever System was used. This measuring system revealed that mechanical stimuli trigger a functional 3D remodeling of skeletal muscle towards a cardiac muscle-like structure. Molecular and functional analyses supported these findings, highlighting the effectiveness of X-MET to express relevant biomarkers of functional cardiomyocytes in comparison to an unstimulated and 2D skeletal muscle culture system. This technology shows promising results that could aid in development of new strategies for regenerative medicine.
Bioengineered 3D Skeletal Muscle Constructs for Aging Studies
A thorough mechanistic understanding of cell-autonomous changes in skeletal muscle after injury is particularly important for aged populations, as this could lead to novel interventions resulting in improved functional recovery. However, due to the limitations of traditional 2D biological aging models, major knowledge gaps persist. Wang et al. (2023) made use of a 3D muscle aging system to characterize the effects from non-muscle cells and other organs on aging mammalian skeletal muscle.
Through the use of this system, as well as the 405C Force Transducer, 322C High-Speed Length Controller, and the 600A Real-Time Muscle Data Acquisition and Analysis Software, Wang et al. (2023) demonstrated that aged/old muscle constructs present a sarcopenic phenotype. This is supported by hypotrophic myotubes, reduced contractile function, and decreased regenerative capacity of old muscle constructs compared to young ones. These old constructs also highlight the regenerative responses of aged muscles in response to 3 pharmaceutical and biological interventions. Additionally, analyzing muscle-cell specific mechanisms that could contribute to impaired regeneration over time revealed that aging induced an increase in complement component 4b (c4b), delaying muscle progenitor cell amplification, and impairing functional recovery. Muscle regeneration in vitro and in vivo was improved by administering a c4b inactivator, known as complement factor 1, suggesting that c4b inhibition may be a beneficial approach for aged muscle repair.
Conclusions
These research endeavors illustrate the sophisticated techniques and innovative processes that are currently shaping the field of regenerative medicine. The utilization of Aurora Scientific’s precise instruments has reaffirmed the role of advanced research tools in driving forward scientific exploration and therapeutic innovation. Together, these studies highlight promising avenues for the development of novel regenerative therapies, guided by an improved understanding of cellular and tissue mechanics.



