In the fields of muscle physiology and neuroscience there is often a lack of overlap in experimental focus even though it is known that the nervous system and peripheral tissues like muscle are not mutually exclusive. Muscle physiologists mainly focus on what happens with the muscle both structurally and mechanistically often overlooking what happens presynaptically and postsynaptically in motor neurons and sensory afferents. In contrast, neuroscientists study the nervous system output and input often disregarding what is happening downstream in peripheral tissues like muscle.
However, in neurophysiology the communication between the nervous system and peripheral tissues like muscle for example, is paramount to understanding how contractions are initiated at the neuromuscular junction as well as the sensory feedback that is relayed to the central nervous system through afferents. In this blog we will outline areas of interest within neurophysiology where our equipment can be used to elucidate the mechanical and physiological properties of proprioception, motor neuron regeneration and neuromuscular junction function and regeneration in models of aging, mutation and disease.
Proprioception
Motor Neuron Function and Regeneration
Neuromuscular Junction Function and Regeneration
Proprioception
Proprioception is how the nervous system senses our body in space, in the field of neurophysiology it consists of sensory afferents sending feedback from the muscle in the periphery to the central nervous system to convey information about our environment such as uneven ground or in some cases disease states that affect function. In this field, researchers study how contraction or stretch of muscle activates these neuronal afferents to ascertain this information.
Dr. Katherine Wilkinson at San Jose State University studies proprioception using isolation of the extensor digitorum longus (EDL) muscle with attached nerve to test afferent responses to muscle stretch in the adult mouse. Using our 300C Dual-Mode Muscle Lever System mounted on our 809C-IV in-vitro muscle bath she is able to simultaneously stretch the EDL while recording sensory activity with a suction electrode. With this setup, researchers are able to study many other types of muscle afferents as well as their response to drug application and disease states using various genetic approaches.
Figure 1. Image of 809C-IV in-vitro test apparatus with intact EDL nerve-muscle preparation for measuring stretch-activated firing of sensory afferents. Adapted from Franco et al. JoVE (2014).
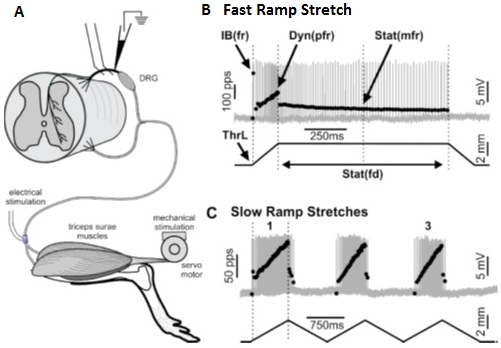
Although Dr. Wilkinson performs this research using isolated muscle with an intact sensory nerve, there are also cases where other techniques can be used as shown in this paper by Vincent et al. (2017). Here, mechanosensory responses were recorded in an in-situ preparation whereby the hindlimb, sensory nerves and spinal cord were kept intact to allow for measurement of this proprioceptive feedback within the dorsal root. Using our 300C Dual-Mode system, the triceps surae muscle of the rat was passively stretched and this mechanical stimulation was recorded from the intraspinal destinations of these muscle proprioceptors. With this assay, firing properties in response to slow or fast stretching can be measured and ultimately help decipher detriments incurred in various disease states.
>> View JOVE
Figure 2. Diagram depicting in-situ preparation for recording action potentials in the dorsal root in response to stretch of triceps surae via Achilles tendon. Adapted from Vincent et al. J. Neurophysiol (2017).
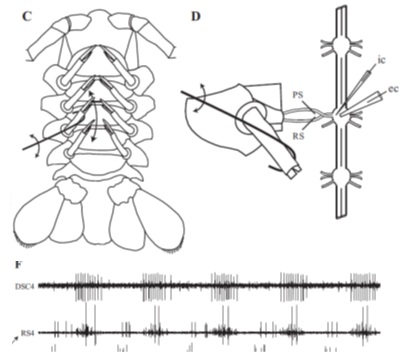
This type of sensory feedback can also be tested in invertebrate models such as the crayfish or moth. In crayfish, researchers utilized a 322C High-Speed Length Controller to retract or protract swimmerets while recording from the associated intact ganglion. Sensory information like this can be used to determine how environmental input during swimming can alter direction or speed of the animal.
Figure 3. Figure illustrating hook attached to length controller used to retract crayfish swimmeret (C and D). F: recording from intact ganglion in response to retraction and protraction. Adapted from Mulloney et al. J. Neurophysiol. 2014.
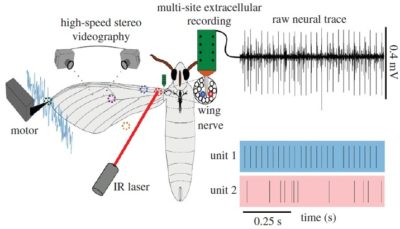
Similarly, Tom Daniel’s group at the University of Washington set out to test whether the hawkmoth wing sensilla are capable of encoding mechanosensory information in response to mechanical stimuli elicited with our 305C Dual-Mode lever system. This was combined with novel techniques using infrared laser and high-speed video while simultaneously recording from the wing nerve to uncover this multi-sensory role.
Figure 4. Experimental set-up for mechanically stimulating the hawkmoth wing campaniform sensilla to identify their mechanosensory properties along with representative neural recordings. Adapted from Pratt et al. Proc R Soc B Biol Sci (2017).
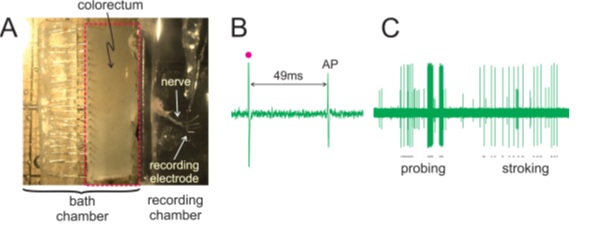
In addition to studying afferents in skeletal muscle using hindlimb muscle or diaphragm strips, there are many researchers looking at stretch-activated afferents in smooth muscle such as from the stomach or in Dr. Bin Feng’s case, the colon. Using an intact nerve-muscle preparation of a dissected colorectum and pelvic nerve, Dr. Feng can study how various mechanical perturbations such as probing, stroking or in the case of our 300C Dual-Mode, stretching, can affect sensory afferent firing properties.
While simultaneously recording from the pelvic nerve, you can ascertain real-time afferent responses to computer controlled stretching of the colorectum tissue. In fact, Dr. Feng has recently been testing how simultaneous bidirectional stretching of the muscle alters the firing properties of these nerves with our 2-Channel Dual-Mode lever system (300D).
Figure 5. Image of the dissected colorectum with attached pelvic nerve (A) and representative recordings of an action potential elicited via electrical stimulation (B) and representative neuronal responses to monofilament probing and mucosal stroking (C). Adapted from Feng et al. JoVE (2015).
>> View JOVE
Motor Neuron Function and Regeneration
Whether it be through neuropathy, injury, age-related loss or in many cases, neurodegenerative disease like amyotrophic lateral sclerosis (ALS) or spinal muscular atrophy (SMA), the ability to regenerate damaged or lost motor neurons is imperative to slowing or reversing the progression of these disease states. Regenerating these connections takes a long period of time and often fails due to the distance required for reconnection. Therefore the ability to measure neuromuscular function before and after treatment in-vivo is paramount to determining the level of functional recovery in these animal models over days, weeks and months of treatment.
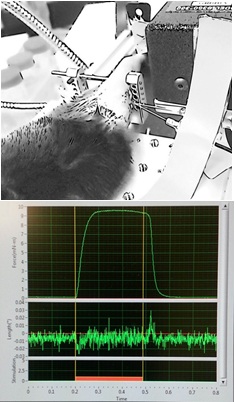
In the case of Dr. Young-Jin Son’s research at Temple University, the aim is to promote long-distance regeneration and enable these motor nerves to re-innervate their peripheral targets, such as muscles. His lab has shown significant peripheral nerve regeneration by preventing or reversing the atrophy of Schwann cells, which over time, lose the ability to support axon regeneration following injury. Utilizing this approach, Dr. Son is employing our 1300A 3-in-1 Whole Animal System to measure recovery of hindlimb muscle function in mouse models of peripheral neuropathy and neuromuscular disease.
Figure 6. Direct sciatic nerve stimulation measuring plantarflexion in-vivo (Top) and software screenshot of a tetanic contraction of the mouse plantar flexors in-vivo (Bottom). Courtesy Dr. Harun Noristani, Dr. Young-Jin Son Lab, Shriners Pediatrics Research Center.
Using this system, the behavioral aspect of functional assays like treadmill running, grip test or Rota Rod is eliminated and muscle function can be characterized by parameters such as maximum tetanic force, fatigability and motor unit number.
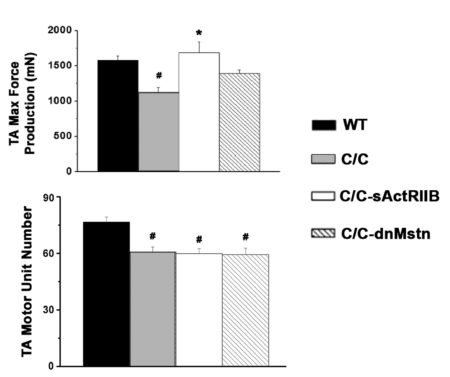
In the case of Lee Sweeney’s lab at the University of Florida, motor unit number and maximum force were measured in-situ to determine muscle specific effects of Activin receptor type IIB (ActRIIB) or myostatin inhibition in a mouse model of spinal muscular atrophy.
Figure 7. Max force and motor unit number measured in-situ from Tibialis Anterior in wildtype (WT), SMAc/c control and SMAc/c mice treated with ActRIIB or Myostatin. Adapted from Liu et al. PLoS One (2016).
Neuromuscular Junction Function and Regeneration
Not only do motor neurons need to regenerate, they must also form functional neuromuscular junctions (NMJs) to not only survive but also activate the muscle thereby reducing atrophy and promoting recovery in various disease states. Studying the functionality and efficacy of the NMJ can be done in-vivo, in-situ and in-vitro by measuring the contractility of the muscle. The involvement of various proteins in forming and maintaining the NMJ can be tested by genetically altering their expression in animal models and measuring the strength of the NMJ in response.
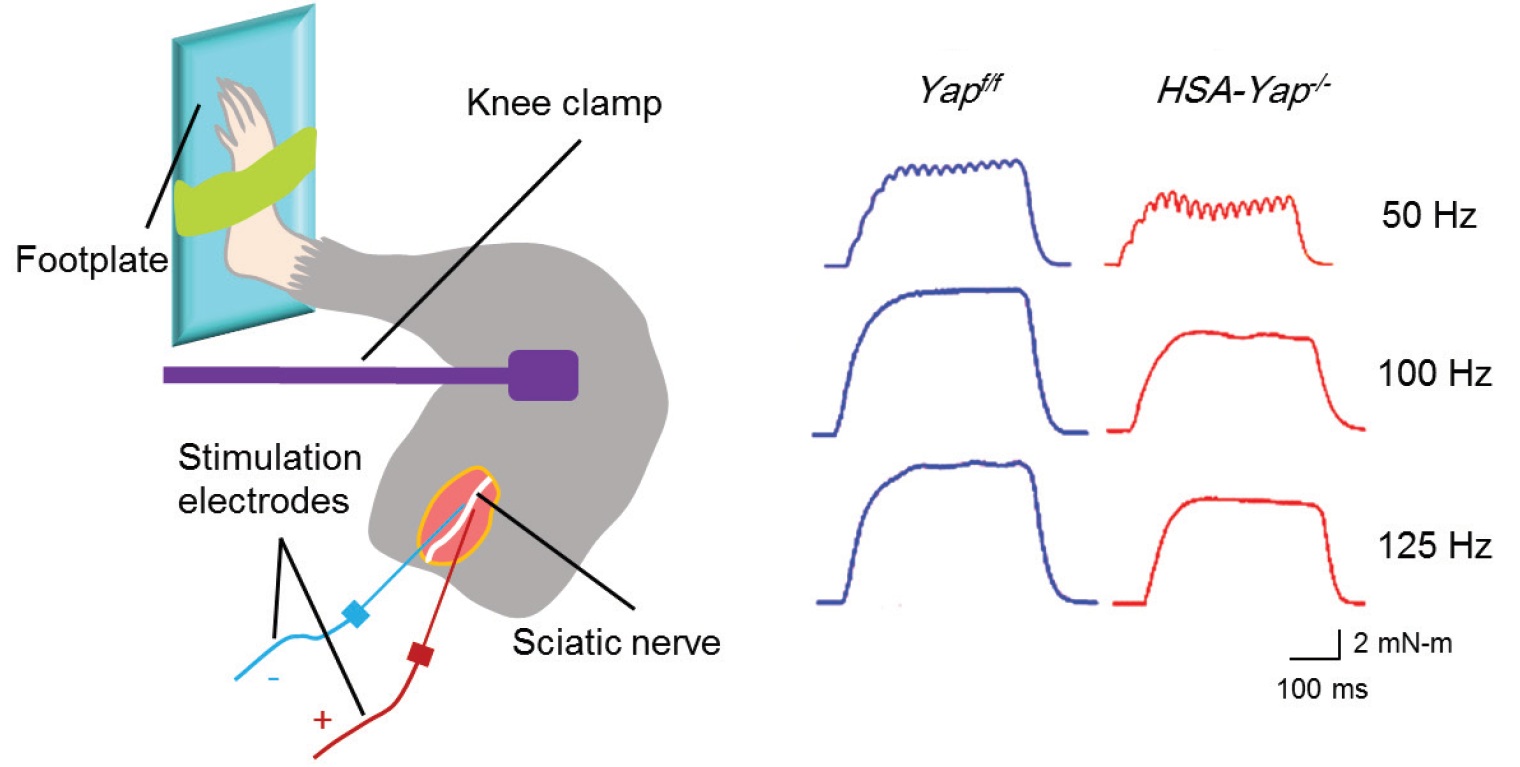
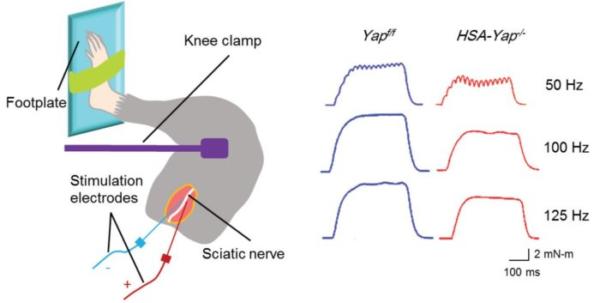
One particular protein, the Yes-associated protein (Yap), is of interest to Dr. Lin Mei’s lab at Augusta University and they have employed our 1300A in-vivo muscle system to measure hind limb muscle function in response to sciatic nerve stimulation with or without Yap. The effects of this knockout can clearly be shown in the resultant tetanic contractions (Figure 8).
Figure 8. Schematic depicting in-vivo muscle twitch and tetanus measurement via sciatic nerve stimulation and representative tetanic curves at stimulation frequencies of 50Hz, 100Hz, 150Hz. Adapted from Zhao et al. J Neurosci (2017).
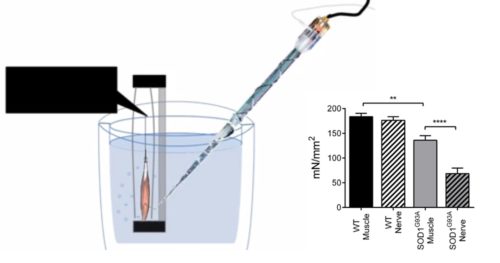
Furthermore, experimentally measuring NMJ functionality can be done by comparing the contractile response to direct muscle or nerve stimulation in an intact muscle-nerve preparation using our 1200A Isolated Muscle System. By doing this, Dr. Emmanuele Rizzuto et al. have elucidated changes in the NMJ that result in a functional deficit in the SOD1G93A mouse model of ALS.
Figure 9. Specific tetanic force measured from soleus intact nerve-muscle preparation. Adapted from Rizzuto et al. JoVE (2017).
>> View Jove
Although studying the neuromuscular junction in vertebrates is incredibly useful, the power of genetics in the Drosophila provides researchers in the Littleton Lab at MIT the ability to generate numerous fly lines to aid in studying how synaptic proteins regulate vesicular trafficking in presynaptic neurons. Recently the lab has begun looking at excitation-contraction coupling and how critical proteins regulate or alter the efficacy of the neuromuscular junction. Utilizing genetically altered fly lines, you can measure the effects of these proteins on whole larval body contractions which can be measured with our isometric 400A Series Force Transducers as shown in Figure 10.
Figure 10. Whole Drosophila larvae (red arrow) fixed at one end with a minuten pin and the other to a 400A force transducer with custom minuten pin claw to attach to larvae (black arrow) (Top). Representative trace of whole body muscle twitches (bottom). Courtesy of Littleton Lab, MIT.
These are just a few examples of how our equipment can be implemented to uncover mechanisms underlying neuromuscular function in normal and disease states. Although this equipment is primarily implemented in muscle physiology, it is important to realize that muscle is not the beginning and end of the story and that assessing the interplay between muscle and nerve can aid in understanding the entirety of a disease’s affects. If you’d like more information, feel free to contact us to discuss your experimental needs.
Additional Published Examples: