Muscle spindles detect changes in muscle length, which is conveyed to the central nervous system (CNS) through afferent nerve fibers. The CNS uses this stretch information to compute limb position and movement, which is necessary for stable motor control. Although spindle function is impaired in many neuromuscular diseases, which can result in an unstable gait and frequent falls, muscle spindles are usually overlooked when examining muscle function and designing therapies for these diseases. At Aurora Scientific, we provide instrumentation that can facilitate the study of sensory encoding by muscle spindle afferents, which we highlight in this publication review.
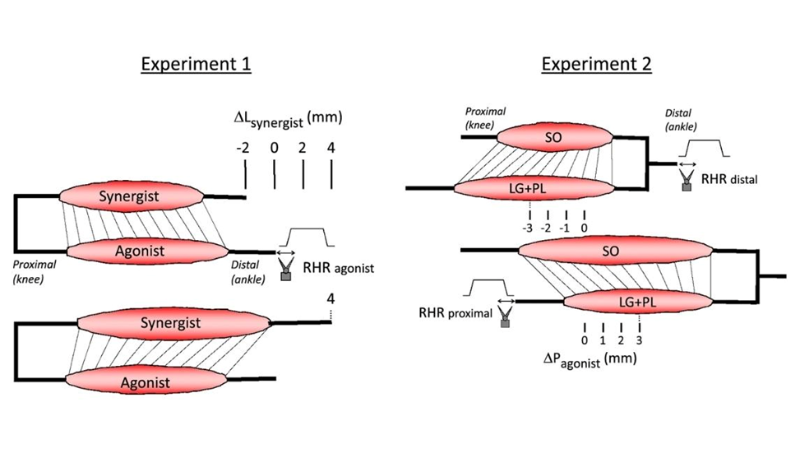
Featured image (© 2022 Maas et al., licensed under CC BY 4.0) illustrates muscle lengths and relative positions for each experimental protocol in the study.
Vesicle-Released Glutamate is Necessary to Maintain Muscle Spindle Afferent Excitability but Not Dynamic Sensitivity in Adult Mice
The muscle spindle is innervated by afferents that constantly report muscle length and movement. Although the PIEZO2 mechanically-sensitive cation channel is known to be essential for stretch sensitivity, the mechanism by which these afferents translate muscle movement into action potential is poorly understood. Previous studies have reported that glutamate can increase spindle afferent firing during static stretch, while glutamate antagonists can attenuate firing. Recently, Than et al. examined whether vesicle-released glutamate is necessary for maintaining muscle spindle afferent excitability during static and dynamic muscle stretching, and employed our 300C-LR Dual-Mode Muscle Lever system and 701C High-Power Stimulator.
The authors found that glutamate significantly increased the spindle afferent firing rate during the plateau phase of stretch, but not during the dynamic phase. Furthermore, blocking glutamate packaging into vesicles both pharmacologically and transgenically decreased firing during sustained stretch. The authors conclude that vesicle-released glutamate is necessary for maintaining muscle spindle afferent excitability, but not for dynamic sensitivity, and note that the importance of afferent-released glutamate in the development of muscle pain conditions should be examined in the future. Investigating this underlying signalling pathway can not only aid in better understanding mechanosensation in spindle afferents, but can also help define therapeutic targets for diseases with abnormal afferent excitability.
Effects of Thrust Magnitude and Duration on Immediate Post-Spinal Manipulation Trunk Muscle Spindle Responses
Pharmacological management for chronic lower back pain, which affects more than 500 million individuals worldwide, has limited success and can be highly addictive. A common non-pharmacological therapy for lower back pain is high velocity low amplitude spinal manipulation (HVLA-SM), but its underlying neurophysiological mechanisms are unknown. While trunk muscle spindle responses during HVLA-SM thrust delivery have been well-characterized, little attention has been paid to muscle spindle discharge recovery after peak force delivery. Therefore, Lima et al. recently investigated trunk muscle spindle responses immediately following HVLA-SM at various thrust magnitudes and durations through secondary analysis of data collected from a feline model. Simulated HVLA-SM thrusts were delivered using our 310C Dual-Mode Lever system.
The authors found an association between thrust magnitude and immediate muscle spindle response, with 55% feline body weight thrust magnitude having the most consistent impact on immediate post-discharge outcomes. However, no association was observed between thrust duration and immediate muscle spindle response. The authors note that future studies should look into HVLA-SM delivery in inflammatory or chemosensitized tissue environments to greater understand its immediate and long-term effects on muscle spindle responses in the context of lower back pain.
Detection of Epimuscular Myofascial Forces by Golgi Tendon Organs
Skeletal muscles contain many tendon organs that are located at both the proximal and distal ends of muscle fibers, but the reason why they are distributed in this manner is unknown. Thus, Maas et al. investigated how mechanical interactions between adjacent muscles affect sensory encoding by tendon organs in Wistar rats. In one experiment, the authors assessed how gastrocnemius and plantaris (GAS + PL) or soleus (SO) muscle stretching impacts tendon organ firing behavior; the distal tendons of GAS + PL and SO were attached to our 309C and 305L-BR lever systems, respectively. In another experiment, the authors imposed physiological changes in muscle length and relative position, this time investigating lateral gastrocnemius and plantaris (LG + PL) muscles in addition to SO muscles. The distal tendons of LG + PL + SO and the proximal tendons of LG + PL were attached to separate 309C servomotors.
Lengthening GAS + PL was found to decrease the force threshold of SO tendon organs. In contrast, SO length did not affect the force threshold of LG tendon organs. Additionally, displacing LG + PL from a proximal to distal position decreased the force threshold of LG tendon organs, indicating that changes in length and/or relative position of adjacent synergistic muscles affects tendon organ firing. The authors conclude that tendon organs can provide information about local stresses caused by epimuscular myofascial forces to the CNS, but note that further studies should look into if and how their anatomical organization is used to control movement.