Image credits here…
Neuromuscular disorders can have severe symptoms that effect the ability to complete even simple daily activities. While their root causes vary widely, excessive muscle dysfunction is a common symptom for many of these disorders. When muscle dysfunction results in hyperactivity, it can adversely affect quality of life. Spasticity in skeletal muscle can impair precise movements and locomotion, while hyperactivity in smooth muscle can lead to incontinence. When muscle dysfunction results in inactivity and paralysis, locomotion and other movements can become completely impossible, resulting in a loss of independence.
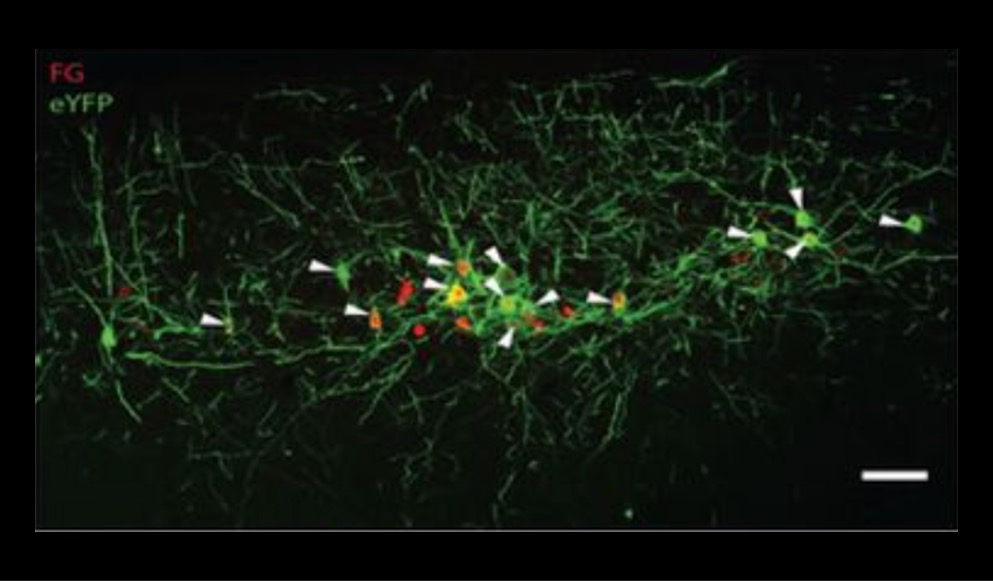
“New developments in this field are now attempting to use targeted light as a way to control muscle activity. Optogenetics uses light-activated proteins, called opsins, to control the stimulation or inhibition of muscles.”
Neuromuscular dysfunction research often involves electrical stimulation of the muscles to invoke contraction. However, invasive implantation of electrodes and continued use of electrical stimulation can cause tissue damage and inhibit normal muscle function. New developments in this field are now attempting to use targeted light as a way to control muscle activity. Optogenetics uses light-activated proteins, called opsins, to control the stimulation or inhibition of muscles. This can be done ex vivo or in vivo, however both methods involve targeted illumination with specific wavelengths of light to activate these opsin proteins. This method allows researchers to non-invasively control muscles without the need for implantation of stimulation electrodes.
The following two publications exhibit the use of optogenetics in two ways: E. Ganji et al. used light-activated opsins to stimulate muscles to produce sustained contraction, while C. Gorini et al. used optogenetics to inhibit muscle contraction for reducing muscle hyperactivity.
Optogenetic activation of muscle contraction in vivo
The aim of this study was to evaluate stimulation feasibility and sustain isometric muscle contraction for an extended period of time using non-invasive, transdermal light activation of skeletal muscle in vivo. LED light was used, through the skin, to stimulate muscle contractions in the hindlimb at differing frequencies to generate a prolonged contraction with the greatest force.
They used Aurora Scientific’s 1300A Whole Animals System to monitor the force of contraction produced with each stimulation pattern and frequency to determine the pattern that produced the strongest contractions. The results of this study demonstrate the feasibility and repeatability of non-invasive optogenetic stimulation of muscle in vivo and highlight optogenetic stimulation as a powerful tool to directly activate skeletal muscle.
Reversible temporally-specific inhibition of muscle using a light-activated chloride channel
In this study, recent innovations in opsin engineering were examined to determine virally mediated, temporally specific optogenetic inhibition of motor neurons and muscle activity in vivo. Optical excitation during tetanic electrical stimulation of skeletal muscle in wild-type mice resulted in inhibition of muscle activity compared to tetanic electrical stimulation without illumination.
Force of contraction was measured using our 300C-LR dual-mode lever system. The extent of optogenetic inhibition in this study was proportional to illumination intensity, where increased light intensity caused further inhibition of muscle activity. These results could have therapeutic applications for neuromuscular disorders that exhibit symptoms related to muscle hyperactivity, and could also provide a new tool to modulate motor neuron activity for in vivo neuroscience and muscle physiology research