In addition to our muscle function and physiology products, we offer solutions for olfaction experiments that have been successfully used in studies involving mice, rats, moths, mosquitos, and ants. This publication review takes a look at two recent studies that examined the mouse olfactory bulb with help from our olfaction instruments.
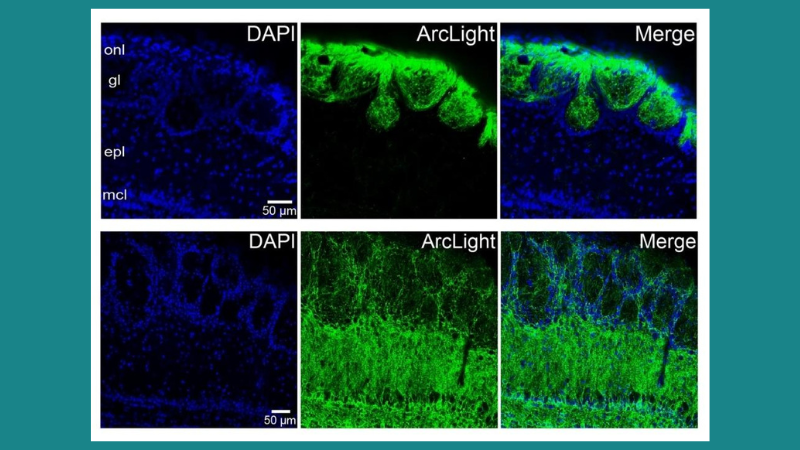
Featured image (© 2022, Platisa, J. et al., licensed under CC BY 4.0) highlights that expression of ArcLight in the olfactory bulb of transgenic mice is cell specific.
Flexible Categorization in the Mouse Olfactory Bulb
In olfaction, categorical representation of odors has been observed in the olfactory bulb and has been described in mitral cells as sudden transitions in responses to odors. Since it is unknown to what extent these response dynamics reflect perceptual categories, Kudryavitskaya et al. examined how learning affects category formation in the mouse olfactory bulb. Mitral cell responses in mice were imaged as they learned two odor classification tasks with different logic. In these tasks, the mice learned to classify odor mixtures based on the dominant compound, as well as how to classify odors independent of their chemical similarity. The authors used our 200B miniPID Fast Response Olfaction Sensor to calibrate the final odorant concentration that was delivered to the animals. Data from this study demonstrates that odor representations by mitral cells are influenced by the learning and task, and that they carry category-related information.
Voltage Imaging in the Olfactory Bulb Using Transgenic Mouse Lines Expressing the Genetically Encoded Voltage Indicator ArcLight
Genetically encoded voltage indicators (GEVIs) can report membrane potential changes from defined cell types and have been used to study sensory processing in olfactory bulb cells. Although recent advances have improved GEVI sensitivity, in vivo GEVI experiments would benefit from increased selectivity in specific cell types. In this article, Platisa et al. report a novel transgenic mouse line that can selectively drive expression of the GEVI ArcLight, localized to either olfactory receptor neurons or olfactory bulb interneurons. The authors examined different aspects of olfactory bulb physiology and found that voltage responses were odor-specific and concentration-dependent. For this study, wide-field epifluorescence and two-photon microscopy were used to functionally characterize these mouse lines, while time course and relative odor concentrations were confirmed with our miniPID. The authors concluded that this transgenic line is a flexible tool for recording the neuronal electrical activity of different cell types, as well as one of the few options for using transgenic mice in vivo.