Mitochondria play key roles in skeletal muscle and undergo remodeling in response to various physiological stresses, but the underlying mechanisms have yet to be fully understood. Highlighted in this publication review are systems available from Aurora Scientific that have aided studies of mitochondrial bioenergetics and muscle function in various research areas, including chronic kidney disease (CKD), sarcopenia, and chronopharmacology.
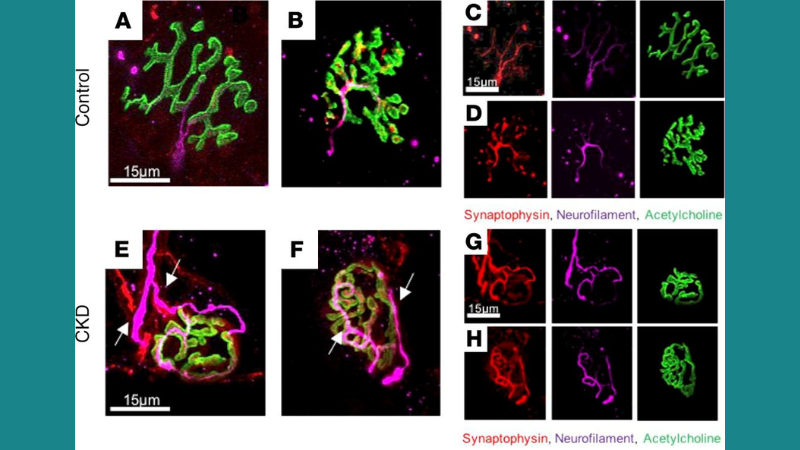
Featured image (© 2021 Thome, T. et al.) consists of immunofluorescence images of neuromuscular junction (NMJ) morphology in the tibialis anterior (TA) muscles of control and CKD mice. Polyinnervation in the NMJ of CKD mice is highlighted by the white arrows, which indicates NMJ remodeling.
Impaired Muscle Mitochondrial Energetics is Associated with Uremic Metabolite Accumulation in Chronic Kidney Disease
There is currently no cure for CKD, which causes progressive skeletal myopathy and inhibits functional independence. Skeletal muscle mitochondria have been implicated as contributors to CKD-associated myopathy, but the underlying molecular mechanisms are unknown. To address this research gap, Thome et al. employed a mitochondrial phenotyping platform which included the assessment of oxidative capacity, mitochondrial hydrogen peroxide (H2O2) production, enzyme activities, muscle protein degradation, and muscle contractile function in a mouse model of CKD. Muscle contractile function was assessed using our 300C-LR Dual-Mode Muscle Lever System, and our 701C Electrical Stimulator was used to deliver stimulation frequencies to measure isometric forces. Mitochondrial oxidative phosphorylation was significantly reduced in these mice, which was determined to be driven by reduced matrix dehydrogenase activity. CKD mice also experienced muscle atrophy and increased muscle protein degradation compared to control groups, and exhibited morphological changes that were indicative of NMJ destabilization. The authors noted that this study provides the first comprehensive evaluation of mitochondrial health in murine CKD muscle.
Scavenging Mitochondrial Hydrogen Peroxide by Peroxiredoxin 3 Overexpression Attenuates Contractile Dysfunction and Muscle Atrophy in a Murine Model of Accelerated Sarcopenia
Sarcopenia, the progressive loss of muscle mass and strength with age, compromises health and quality of life in the elderly. Although the underlying molecular mechanisms are not defined, its known risk factors include excess reactive oxygen species, NMJ disruption, and loss of innervation. To determine how mitochondrial H2O2 affects neurogenic atrophy and contractile dysfunction, Ahn et al. studied mice with muscle-specific overexpression of the mitochondrial H2O2 scavenger peroxiredoxin3 (mPRDX3) that were crossed with SOD1 knockout (KO) mice, an established model of sarcopenia. Mouse muscle contractile properties were investigated using our Dual-Mode Muscle Lever System, while other experiments included NMJ imaging, mitochondrial functional assays, enzyme and protein analyses, and immunohistochemistry. Overexpression of mPRDX3 was found to normalize elevated rates of H2O2 generation in Sod1KO mouse muscle mitochondria, prevent declines in the maximum mitochondrial oxygen consumption rate and calcium retention capacity, mitigate muscle atrophy, and preserve maximum isometric specific force at wildtype (WT) levels. However, mPRDX3 overexpression could not resolve NMJ disruption. The authors concluded that mPRDX3 overexpression improves mitochondrial function and mitigates muscle loss despite persistent NMJ impairment, which can help narrow down drug targets for sarcopenia treatment.
Muscle Mitochondrial Remodeling by Intermittent Glucocorticoid Drugs Requires an Intact Circadian Clock and Muscle PGC1α
Glucocorticoids are naturally-secreted steroid hormones with a prominent circadian rhythm, and exogenous glucocorticoids like prednisone are widely used as anti-inflammatory drugs. Intermittent dosing of prednisone once per week has been reported to elicit distinct effects in mice with muscular dystrophy compared to the common once-daily dosing regime. Since the muscle-autonomous metabolic and circadian determinants of glucocorticoid pharmacology are unknown in both normal and diseased muscle, Quattrocelli et al. investigated how dosage time impacts the effects of prednisone on muscle bioenergetics using Bmal1-KO and WT mice. In-situ tetanic force from TA muscle was measured using our 1300A Whole Animal System along with our 300C-LR Dual-Mode Muscle Lever System. Additional experiments included tissue respirometry and muscle metabolite quantification, as well as mitochondrial density, NAD+-ATP, corticosterone, and luciferase assays. Based on results from these experiments, the authors concluded that glucocorticoid chronopharmacology for muscle bioenergetics requires an intact circadian clock as well as muscle PGC1α activity. The authors also noted that the molecular and metabolic effects reported in this study will pave the way for muscle-tailored therapies without off-target drug effects.