It’s no secret that exercise is good for you. Globally, the medical burden of obesity, cardiovascular disease and type-2 diabetes are astronomical, but exercise represents a crucial means of treatment and prevention. However, some muscle pathologies such as severe burns and volumetric muscle loss can result in a permanent loss of muscle function, precluding one’s ability to effectively exercise.
Although treatment for these conditions remains difficult, clever experimental design and effective tools to assess muscle function lead to promising new therapeutic approaches. In the cases below, Southern et al. and Song et al. used the Aurora Scientific 300C-series dual-mode muscle levers to show how exercise can improve muscle function as a treatment or recovery protocol for different types of traumatic muscle injury.
Insulin and exercise improved muscle function in rats with severe burns and hindlimb unloading
Following severe burn, muscle loss due to protein degradation can greatly affect a patients’ quality of life. Complicating matters is the fact that severe burn patients are often immobilized for an extended period of time during recovery resulting in further muscle atrophy and a reduction in muscle mass. And although current treatment often focuses on stabilizing the injury condition, the publication’s authors note that,
“Ultimately, immobilization complicates recovery from injury as much as the original injury itself.”
Because protein degradation is the major driver in muscle loss after burn, exercise could help offset protein turnover by increasing synthesis and breakdown. On a molecular level, exercise increases protein synthesis by activating the Akt-mTOR-p70S6k pathway. Insulin has also been used to help treat burns, as it is known to increase protein synthesis and decrease protein degradation. However, its role in modulating the AKt-mTOR-p70S6k pathway is not fully understood.
In this paper, Song et al. set out to investigate whether a combination of exercise along with the administration of insulin could improve muscle function in a rodent model of burn with subsequent immobilization simulating bed rest.
To do this, they used a rat model of thermal injury with immobilization via hindlimb unloading. They randomly assigned the rodents one of 4 treatment groups: resistance exercise alone, insulin alone, both exercise and insulin, or no treatment. After 14 days, they assessed muscle function using the Aurora Scientific 1305A in-situ system with Dynamic Muscle Control and Analysis software. They found that exercise alone markedly improved function, and exercise and insulin together had an even greater effect on muscle function. These animals demonstrated a comparatively greater tetanic and isometric twitch force.
Following assessments of function, molecular analysis was used to better understand the mechanisms and pathways responsible for these physiological changes. They found that exercise combined with insulin increased levels of phosphorylated Akt, a major player responsible for regulating the cell cycle and protein synthesis.
The authors conclude that insulin combined with exercise has an additive in recovering muscle function following burn injury in rodent models with hindlimb unloading. This research could represent a promising avenue for clinical treatment protocols for burn moving forward.
PGC-1α overexpression partially rescues impaired oxidative and contractile pathophysiology following volumetric muscle loss injury
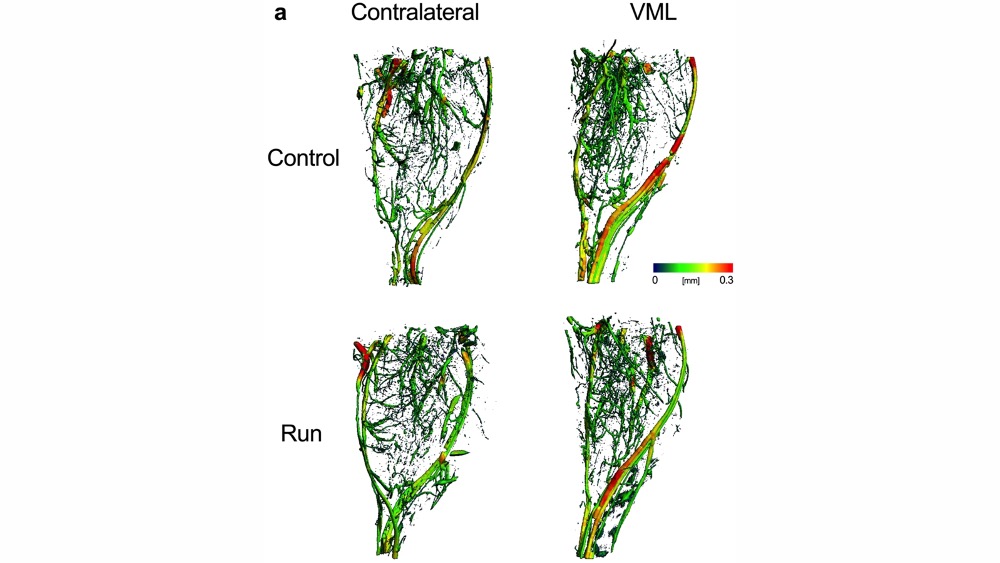
Severe skeletal muscle trauma leading to volumetric muscle loss (VML) can result in permanent disability as the body is not able to regenerate muscle tissue that was lost. Historically, research into VML has focused on histological and contractile properties, but oxidative capacity has been largely overlooked.
In this paper, Southern et al. used mitochondrial respirometry, 2-photon microscopy and in vivo muscle function measurement to assess the impact of VML on oxidative adaptation. In addition, they identified PCG-1α as the limiting factor to exercise-induced adaptation and that overexpression of PCG-1α can substantially rescue muscle function.
To do this, the authors used a mouse model of VML causing chronic impairment of function. Using high-resolution respirometry, they found that the mitochondrial function of remaining muscle was reduced by about 60%, a disproportionate amount compared to the volume of muscle tissue that was lost. They also had mice exercise in the form of a voluntary running wheel. They found that mice with VML demonstrated no oxidative adaptation following the exercise program. To determine whether exercise improved contractile properties in mice with VML, they measured peak isometric torque of plantar flexor muscles using the Aurora Scientific 300C-LR muscle lever. Indeed, contractile properties were improved with exercise independent of any improvement in oxidative capacity, indicating that VML impairs oxidative plasticity but not contractile plasticity.
To test whether transcription factor PGC-1α was a limiting factor to mitochondrial biogenesis following VML, they used a genetic mouse model overexpressing PCG-1α. Four weeks following VML injury, these transgenic mice had 25-35% greater mitochondrial function and improved torque production and contractile fatigue compared to control mice. Overexpression of PCG-1α restored functional capacity and oxidative adaptation.
The authors write,
“ are promising in that VML injured muscle was capable of oxidative adaptations and suggests that mitochondria may play an important and unaddressed role in the recovery of muscle strength after VML.”
They conclude that a treatment strategy including restoration of mitochondrial biogenesis—and by extension muscle function—may greatly benefit patients with VML injury. It’s worth noting that in this case, exercise did not improve all facets of exercise adaptation. Oxidative and metabolic capacity remained impaired, but muscle function itself did dramatically improve following injury.