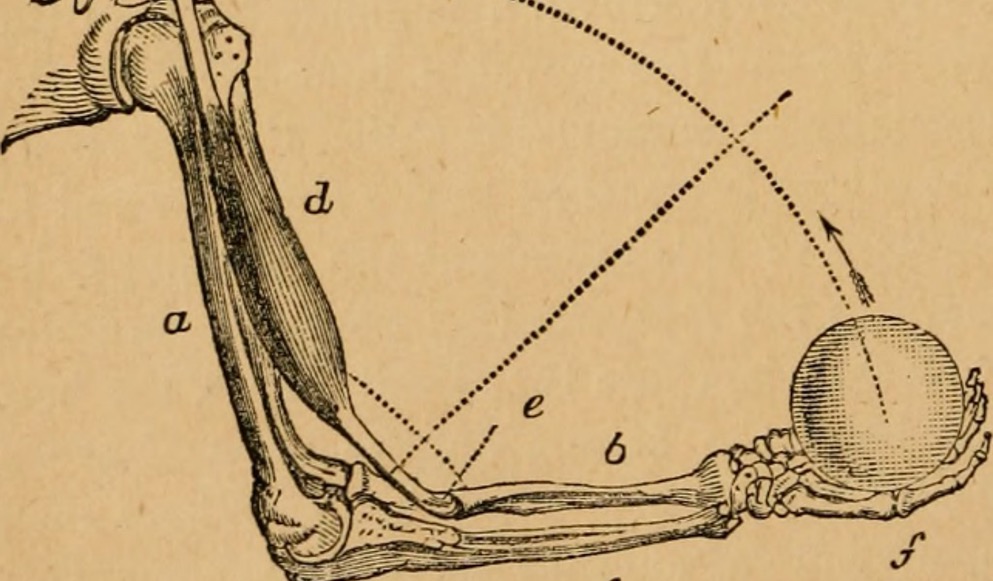
The bone-muscle interface can be thought of as a lever system: the joint acts as the fulcrum while the muscle exerts force to the bone to provide power. However, the short distance between where the muscle attaches and the joint puts the lever at a mechanical disadvantage, and the muscle has to apply considerable force on the bone to achieve an effect. While it’s an inneficient setup in terms of effort, the mechanical force that muscle exerts on bone is critical for bone formation and health.
Similarly to muscle, bones strengthen the more they are used. Consequently, weight-bearing and resistance exercises are an excellent way to increase your bone density and mass. However, if bones are not challenged by regular use, they can become frail. This is of particular concern to individuals with muscle weakness, whether due to age or musculoskeletal or neuromuscular disorders, for whom weight-bearing exercise can be difficult or impossible.
In the two following publications, the authors approached bone-muscle crosstalk research from different angles: Alycia Berman et al. looked at how much strain in-vivo contractions induced on the bone itself (stimulating bone growth), while Dounia Hamoudi et al. used an in vitro approach to look at how strengthening bone could in turn strengthen muscle.
Muscle contraction induces osteogenic levels of cortical bone strain despite muscle weakness in a mouse model of Osteogenesis Imperfecta
Osteogenesis imperfecta (OI) is a genetic disorder in which a defect in collagen synthesis makes the bones of those affected brittle. Due to the intrinsic link between muscle and bone, individuals with OI often have a reduction in muscle strength according to the severity of the disease. In this article, the authors used the oim/oim mouse model of OI to assess how efficiently muscle weakened by OI can exert strain on the bone in vivo.
To address this question, the team used our 1300A: 3-in-1 Whole Animal System to stimulate contraction of the gastrocnemius and soleus muscles, measuring torque on the ankle using the system’s transducer and bone strain using a strain gauge on the tibia. As expected, they observed significant muscle weakness in the oim/oim mice. These mice had a muscle cross-sectional area about half that of the WT mice and produced on average 77% less torque. However, despite the marked muscle weakness, they found that the tensile strain these mice were able to exert was above the threshold required to stimulate bone formation.
If translatable to humans, these results come as good news. Muscle stimulation, even at its reduced level, is still sufficient to induce bone formation in individuals with OI. However, the authors do note that it may not be easy, writing
“it may require more “effort” to induce a bone formation response in oim/oim than in WT.”
Muscle Weakness and Selective Muscle Atrophy in Osteoprotegerin-Deficient Mice
In contrast to the approach of Berman et al. to look at how muscle function could affect bone, the authors of this paper did the reverse, manipulating key regulators of bone remodeling to investigate their effect on muscle function. The regulators in question are OPG and RANKL; RANKL promotes osteoclast activation and bone resorption, while OPG binds to RANKL to inactivate it.
The authors used an Opg–/– mouse model and assessed muscle function over time. In vivo tests included open field running distance and whole limb grip force, and ex vivo assessement was done using electrical stimulation and the 305B-LR force sensor to measure maximum isometric and maximum specific force.
After 5 months, Opg-deficient mice had greater circulating RANKL, traveled less far during open-field tests and had reduced performance for whole limb grip force tests. In addition, twitch, maximum isometric and specific force production ex vivo were reduced. They showed that while contractile properties of fast-twitch fibers in extensor digitorium longus muscles were reduced, the contractile properties of slow-twitch Sol muscles were not significantly different from WT.
The authors conclude that the deletion of OPG has a great impact on bone and muscle and that their results
“…provide a link between RANKL/OPG imbalance and synchronous bone and muscle dysfunctions.”