March ushered in Brain Awareness Week, a global campaign led by the Dana Foundation that unites universities, research institutions, schools, and advocacy groups around a shared mission: bringing the wonders of neuroscience to the public. Each year, the campaign serves as a timely reminder of just how much neuroscience shapes our everyday lives, and how much remains to be uncovered. This year, olfaction findings are the scenter of attention, as three compelling new papers redefine perspectives on the neuroscience of smell. The following publication review highlights how the brain encodes, calibrates, and flexibly updates its representation of the chemical world.
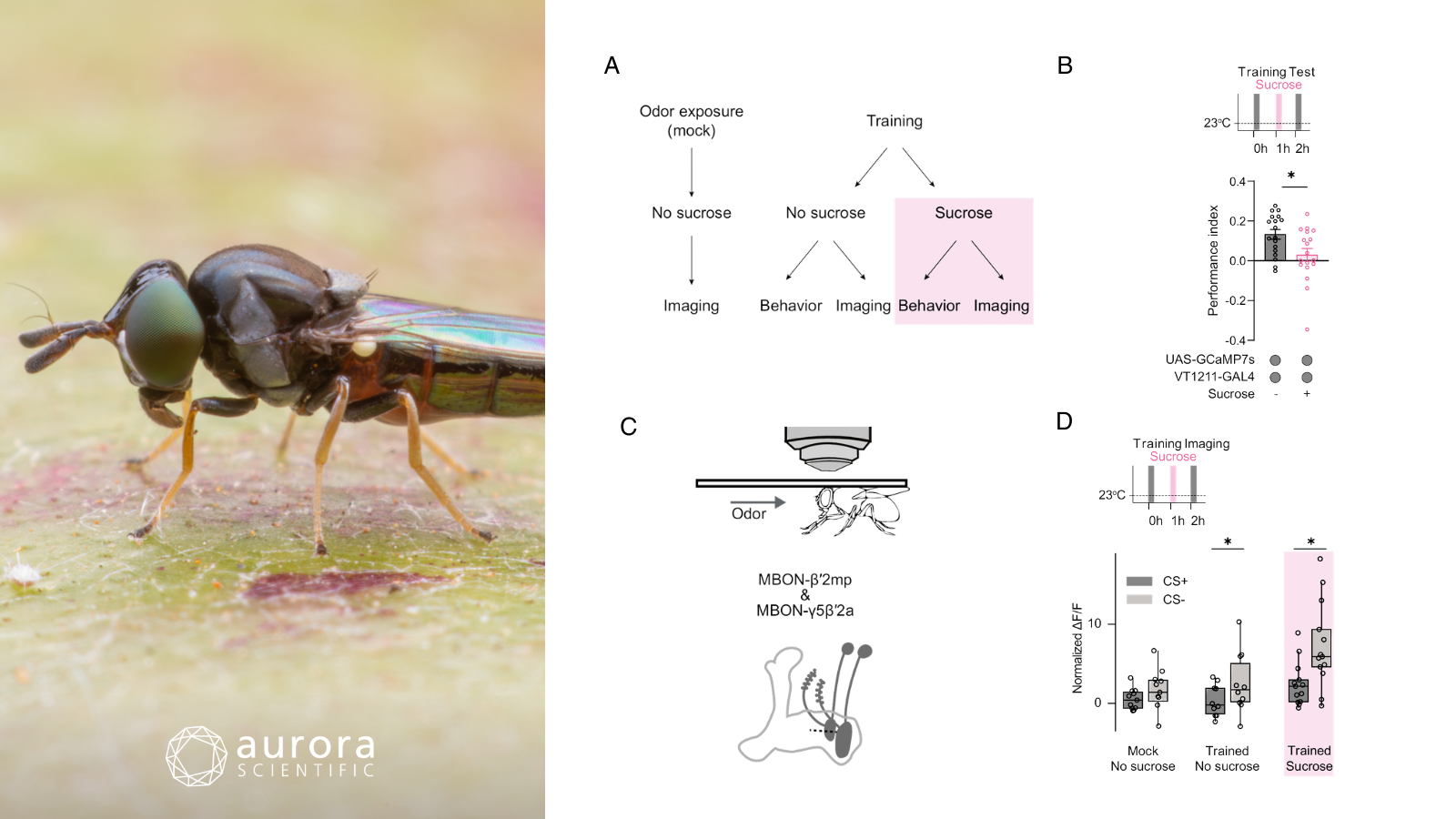
Featured image (photo by piyawat Thongthaengyai from Getty Images, via Canva.com with figures adapted from ©Warnecke et al. (2026), licensed under CC BY 4.0) depicting how memory trace is intact after re-exposure to the unconditioned stimulus. A) experimental procedure, B) sucrose re-exposure 1 hour after learning reduces memory in a 2-hour test (n≥18), C) illustration of the head-fixed two-photon microscope setup (top) and depiction of MBON-β′2mp and MBON-γ5β′2a neurons (bottom), D) calcium responses in MBON-β′2mp and MBON-γ5β′2a at 2 hours post-training with and without the conditioned stimulus (CS) (n≥9, mean ± SEM, individual n indicated by circles, asterisks denote p<0.05, t-test).
Re-exposure to reward re-evaluates related memories
Animals must continually re-evaluate learned associations to adapt their behaviour to changing environments. In classical conditioning, memories are typically modified through re-exposure to predictive cues, yet such approaches often alter only a single cue–outcome association and may not persist across related memories. While re-exposure to an unconditioned stimulus (US) alone has been proposed as a mechanism to broadly re-evaluate associated memories, the conditions and neural processes underlying this effect remain poorly understood. To address this gap, Warnecke et al. (2026) investigated whether re-exposure to a sucrose reward could devalue multiple odour–reward memories in Drosophila melanogaster, and whether this process operated through mechanisms distinct from established dopamine-mediated memory modulation.
Mixed-sex Drosophila were subjected to olfactory appetitive conditioning and tested for odour preference using a T-maze assay. Additional manipulations included optogenetic and thermogenetic neuronal activation, pharmacological inhibition of nitric oxide synthase, and two-photon calcium imaging in head-fixed flies. Aurora Scientific’s 220A: Olfactometer provided precisely controlled airflow (0.9 L/min) and synchronized stimulus delivery, enabling quantification of stimulus-evoked responses in memory-related circuits.
Re-exposure to the sucrose reward after training significantly reduced flies’ learned approach to conditioned odours, demonstrating that reward re-experience can devalue multiple associated olfactory memories even after consolidation. This effect was reward-specific and reflected a reduced attraction to the conditioned odour rather than changes in general motivation or attraction to other cues. Calcium imaging experiments revealed that odour-specific neural responses in mushroom body output neurons persisted after reward re-exposure, indicating that the underlying memory trace remained intact despite reduced behavioural expression. Together, these findings suggest that reward re-experience suppresses the expression of learned associations without erasing the memories themselves, highlighting a mechanism for flexible memory re-evaluation.
A method to estimate absolute odorant concentration of olfactory stimuli
Accurately delivering and quantifying odour stimuli has long been a challenge in olfactory research, particularly in insect sensory physiology where responses strongly depend on stimulus intensity and timing. In many studies, odour stimulus strength is estimated based on the amount of odorant placed in the source, even though evaporation dynamics and airflow can cause the actual delivered concentration to differ substantially. This disconnect limits comparisons across experiments and complicates attempts to reproduce ecologically realistic odour environments. To address this gap, Conchou et al. (2026) developed a mass transfer–based model to estimate the absolute concentration of odorants delivered by a common insect odour delivery system, and applied the approach to examine how the plant volatile (Z)-3-hexenyl acetate influences pheromone detection in Agrotis ipsilon moths.
Air–mineral oil partition coefficients were measured via gas chromatography to model odorant evaporation and movement through the delivery system. Aurora Scientific’s 200B: miniPID measured odorant concentrations at multiple points in the device, validating model predictions and calibrating stimulus intensity. The calibrated system was then applied in electrophysiological recordings examining how defined concentrations of plant volatiles influence pheromone detection in moth antennae.
The model accurately predicted that stimulus intensity depends strongly on each compound’s partition coefficient, with plateau concentrations closely matching predictions. Electrophysiological recordings showed that (Z)-3-hexenyl acetate interfered with pheromone detection only at concentrations unlikely to occur in nature, suggesting limited ecological relevance. These findings provide a practical framework for improving the realism and reproducibility of olfactory experiments.
Odor identity decoding by mitral/tufted cells in the olfactory bulb from large-scale pooled datasets
The olfactory bulb serves as the brain’s first central processing station for smell, where mitral and tufted cells (M/Ts) are responsible for encoding odour identity through specialized neural circuits. While prior work has established that temporal firing patterns, rather than firing rate alone, underlie odour representation, a critical gap remains. Prior studies have relied on relatively small samples of neurons and animals, leaving unresolved how many M/Ts are truly needed for reliable odour decoding and how the number of respiratory cycles influences that accuracy. Li et al. (2026) addressed this by pooling large-scale electrophysiological recordings from 436 M/Ts across 45 mice to interrogate how population size and respiratory dynamics shape the fidelity of odour identity decoding.
Male C57BL/6J mice underwent microelectrode implantation into the mitral cell layer alongside respiratory cannulas to capture sniffing activity. Odour concentration stability was verified using Aurora Scientific’s 200C miniPID. Decoding was performed in Python using support vector machine and minimum distance classifiers with leave-one-out cross-validation.
Upon analysis, odour-evoked firing rate changes were sparse, pointing to temporal firing patterns as the primary coding strategy. Decoding accuracy improved markedly with larger M/T populations, approaching saturation near 150 units, with peak accuracy of ~75%. Performance peaked ~80 ms after inhalation onset and improved further across multiple respiratory cycles, underscoring the role of respiratory-coupled temporal coding in reliable odour representation.
Conclusions
These studies by Warnecke et al. (2026), Li et al. (2026), and Conchou et al. (2026), give a whiff of olfactory processing – from the flexible re-evaluation of odour-reward memories in flies, to the precise quantification of delivered odour stimuli, to the population-level neural codes underlying odour identity in the mammalian brain. Collectively, they underscore how advances in stimulus control, large-scale recording, and behavioural dissection are converging to sharpen our understanding of how the nervous system detects, represents, and adapts to the chemical world around us.